| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
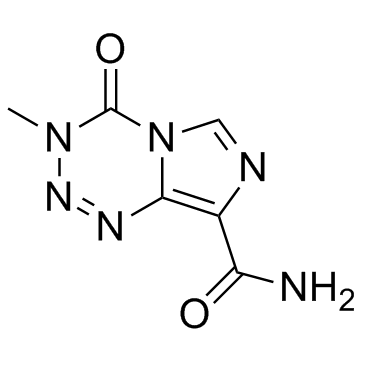 |
替莫唑胺
CAS:85622-93-1 |
|
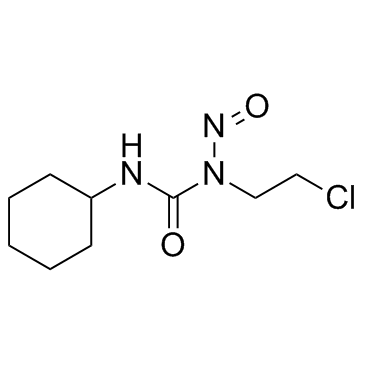 |
洛莫司汀
CAS:13010-47-4 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
替莫唑胺
CAS:85622-93-1 |
|
 |
洛莫司汀
CAS:13010-47-4 |