Isolation, characterization, and nuclease activity of biologically relevant chromium(V) complexes with monosaccharides and model diols. Likely intermediates in chromium-induced cancers.
Ruben Bartholomäus, Jennifer A Irwin, Liwei Shi, Siwaporn Meejoo Smith, Aviva Levina, Peter A Lay
文献索引:Inorg. Chem. 52(8) , 4282-92, (2013)
全文:HTML全文
摘要
The stabilization of Cr(V) by biological 1,2-diolato ligands, including carbohydrates, glycoproteins, and sialic acid derivatives, is likely to play a crucial role in the genotoxicity of Cr(VI) and has also been implicated in the antidiabetic effect of Cr(III). Previously, such complexes have been observed by electron paramagnetic resonance (EPR) spectroscopy in living cells or animals, treated with carcinogenic Cr(VI), as well as in numerous model systems, but attempts to isolate them have been elusive. Recently, the first crystal structure of a Cr(V) complex with cis-1,2-cyclohexanediol (1, a close structural analogue of carbohydrates) has been reported. In this work, Cr(V) complexes of the general formula [Cr(V)OL2](-) [where LH2 = 1, cis-1,2-cyclopentanediol (2), D-glucose (3), D-mannose (4), D-galactose (5), and D-ribose (6)] have been isolated from light-catalyzed reactions of Cr(VI) (anhydrous Na2Cr2O7) with slight molar excesses of the corresponding ligands in N,N-dimethylformamide. The complexes were characterized by elemental analyses, electrospray mass spectrometry (ESMS), and EPR spectroscopy. Studies by electronic absorption spectroscopy have shown that the solids isolated from reactions of Cr(VI) with 3-6 contained mixtures of Cr(V) complexes (40-65 mol %) and Cr(III) species (probably complexes with oxidized ligands), while those from reactions with 1 and 2 were practically pure Cr(V). The first isolation of solids containing significant proportions of chromium(V) monosaccharide complexes led to the definitive assignment of their general formula ([Cr(V)OL2](-), based on ESMS), in agreement with the earlier EPR spectroscopic data. The first direct comparison of the decomposition rates of Cr(V) complexes with 1-6, made possible by isolation of the solids, have shown that the complexes with five-membered-ring ligands (2 and 6) are more stable at pH ∼ 7 compared with their six-membered-ring counterparts (1 and 3-5). This finding emphasizes the likely biological roles of chromium(V) pentose complexes, e.g., those with sugar residues of RNA, ATP, or NAD(P)H. Finally, the first direct evidence for the ability of these Cr(V) complexes to cause oxidative DNA damage in the absence of added reductants or oxidants has been obtained. These data support significant roles for chromium(V) 1,2-diolato complexes in the diverse biological activities of Cr(VI) and Cr(III).
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
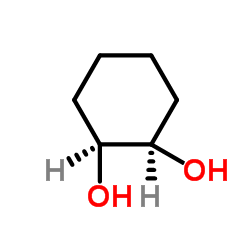 |
顺式-1,2-环己二醇
CAS:1792-81-0 |
C6H12O2 |
|
Effect of ethanol on the urinary excretion of cyclohexanol a...
1999-06-01 [Scand. J. Work. Environ. Health 25(3) , 233-7, (1999)] |
|
Tin(II) chloride catalyzed reactions of diazodiphenylmethane...
2003-04-22 [Carbohydr. Res. 338(9) , 963-8, (2003)] |
|
Biological monitoring of occupational exposure to cyclohexan...
1999-03-01 [Int. Arch. Occup. Environ. Health 72(2) , 115-20, (1999)] |
|
Expression of benzene dioxygenase from Pseudomonas putida ML...
2001-06-01 [Appl. Microbiol. Biotechnol. 55(6) , 721-6, (2001)] |
|
Insight into acid-mediated asymmetric spirocyclization in th...
2000-10-01 [Chem. Pharm. Bull. 48(10) , 1536-40, (2000)] |