Stereoselective total synthesis of bioactive styryllactones (+)-goniofufurone, (+)7-epi-goniofufurone, (+)-goniopypyrone, (+)-goniotriol, (+)-altholactone, and (-)-etharvensin.
Kavirayani R Prasad, Shivajirao L Gholap
文献索引:J. Org. Chem. 73(1) , 2-11, (2008)
全文:HTML全文
摘要
Stereoselective total synthesis of biologically active styryllactones 7-epi-goniofufurone, goniofufurone, goniopypyrone, goniotriol, altholactone, and etharvensin was achieved in high overall yields from a common intermediate derived from d-(-)-tartaric acid. It is based on the utility of a masked tetrol, comprising an alkene tether and four contiguous hydroxy groups. The pivotal reaction sequence involves hydroxy-directed lactonization via the oxidation of alkene, and subsequent elaboration to styryllactones. The masked tetrol was prepared by the extension of gamma-phenyl-gamma-hydroxy butyramide, readily obtained from the bis-dimethylamide of tartaric acid, employing a combination of selective Grignard additions and a stereoselective reduction.
相关化合物
| 结构式 | 名称/CAS号 | 分子式 | 全部文献 |
|---|---|---|---|
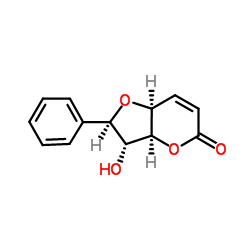 |
Altholactone
CAS:65408-91-5 |
C13H12O4 |
|
The cytotoxicity of naturally occurring styryl lactones.
2006-02-01 [Phytomedicine 13(3) , 181-6, (2006)] |
|
Asymmetric synthesis of (+)-altholactone: a styryllactone is...
2008-01-01 [Chemistry 14(9) , 2842-9, (2008)] |
|
Stereospecificity in the Au-catalysed cyclisation of monoall...
2011-07-21 [Chem. Commun. (Camb.) 47(27) , 7659-61, (2011)] |
|
A convergent Pd-catalyzed asymmetric allylic alkylation of d...
2007-01-01 [Chemistry 13(34) , 9547-60, (2007)] |
|
Altholactone induces apoptotic cell death in human colorecta...
2012-06-01 [Phytother Res. 26(6) , 926-31, (2012)] |