| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
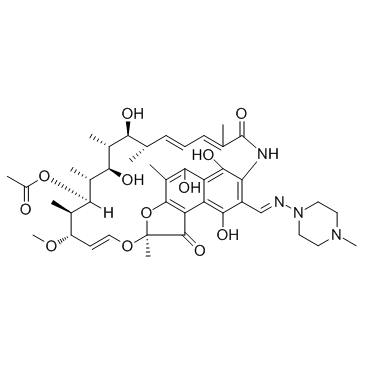 |
利福平
CAS:13292-46-1 |
|
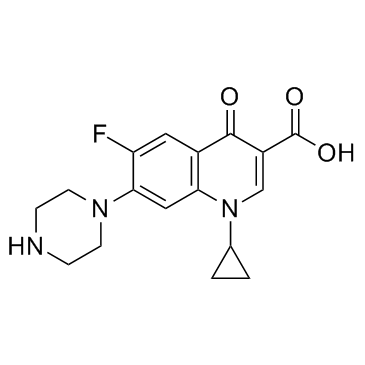 |
环丙沙星
CAS:85721-33-1 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
利福平
CAS:13292-46-1 |
|
 |
环丙沙星
CAS:85721-33-1 |