| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
双酚a
CAS:80-05-7 |
|
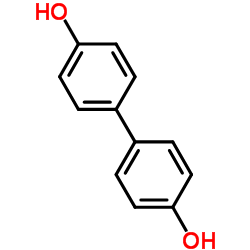 |
4,4'-二羟基联苯
CAS:92-88-6 |
| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
双酚a
CAS:80-05-7 |
|
 |
4,4'-二羟基联苯
CAS:92-88-6 |