| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
硫酸
CAS:7664-93-9 |
|
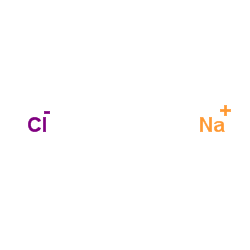 |
氯化钠
CAS:7647-14-5 |
|
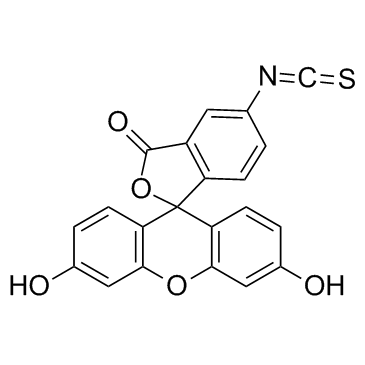 |
异硫氰酸荧光素酯
CAS:3326-32-7 |
|
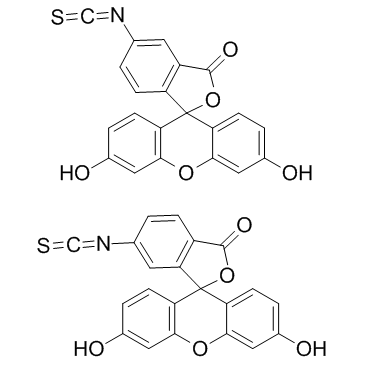 |
异硫氰酸荧光素
CAS:27072-45-3 |
|
 |
钾离子标准溶液
CAS:7440-09-7 |
|
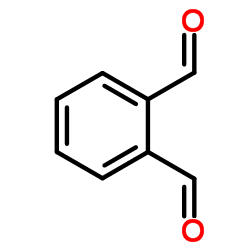 |
邻苯二甲醛
CAS:643-79-8 |
|
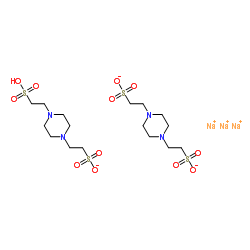 |
哌嗪-1,4-二乙磺酸
CAS:5625-37-6 |
|
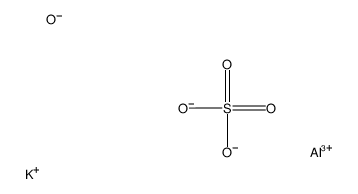 |
明胶
CAS:9000-70-8 |
|
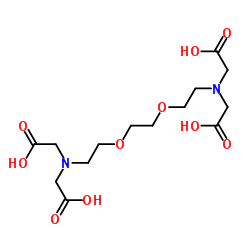 |
3,6-二氧杂-1,8-辛二胺四乙酸(EGTA)
CAS:67-42-5 |
|
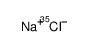 |
氯化钠-35cl
CAS:20510-55-8 |