Synthesis and radiofluorination of novel fluoren-9-one based derivatives for the imaging of α7 nicotinic acetylcholine receptor with PET
Rodrigo Teodoro, Matthias Scheunemann, Barbara Wenzel, Dan Peters, Winnie Deuther-Conrad, Peter Brust
文献索引:10.1016/j.bmcl.2018.03.081
全文:HTML全文
摘要
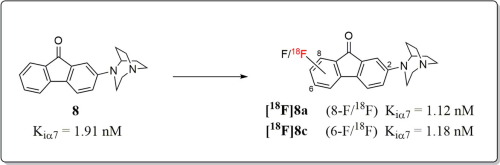
By structure-activity relationship studies on the tilorone scaffold, the ‘one armed’ substituted dibenzothiophenes and the fluoren-9-ones were identified as the most potential α7 nAChR ligands. While the suitability of dibenzothiophene derivatives as PET tracers is recognized, the potential of fluoren-9-ones is insufficiently investigated. We herein report on a series of fluoren-9-one based derivatives targeting α7 nAChR with compounds 8a and 8c possessing the highest affinity and selectivity. Accordingly, with [18F]8a and [18F]8c we designed and initially evaluated the first fluoren-9-one derived α7 nAChR selective PET ligands. A future application of these radioligands is facilitated by the herein presented successful implementation of fully automated radiosynthesis.
|
Design, synthesis, evaluation, and molecular docking of urso...
2018-04-11 [10.1016/j.bmcl.2018.04.021] |
|
Covalent inhibitors of nicotinamide N-methyltransferase (NNM...
2018-04-10 [10.1016/j.bmcl.2018.04.017] |
|
Valproic Acid Induces Three Novel Cytotoxic Secondary Metabo...
2018-04-10 [10.1016/j.bmcl.2018.04.018] |
|
Agonists of the γ-Aminobutyric Acid Type B (GABAB) Receptor ...
2018-04-07 [10.1016/j.bmcl.2018.04.003] |
|
Synthesis of 3-(3-hydroxyphenyl)pyrrolidine dopamine D3 rece...
2018-04-04 [10.1016/j.bmcl.2018.03.084] |