Quinacrine (dihydrochloride)
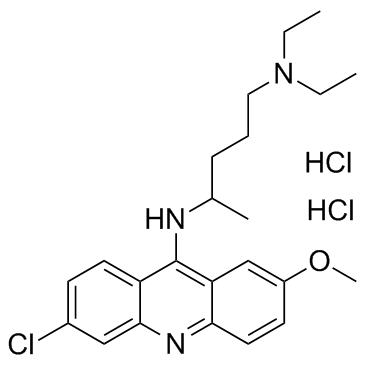
Quinacrine (dihydrochloride) structure
|
Common Name | Quinacrine (dihydrochloride) | ||
|---|---|---|---|---|
| CAS Number | 69-05-6 | Molecular Weight | 472.88 | |
| Density | 1.2962 (rough estimate) | Boiling Point | N/A | |
| Molecular Formula | C23H32Cl3N3O | Melting Point | ca. 248 - 250ºC (decomposes) | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Quinacrine (dihydrochloride)Quinacrine is a fluorescent probe for the conformational transitions of the cholinergic receptor protein. Quinacrine shows activity in the low μM range with a mean IC50 of 2.30 μM In the patient AML cells.IC50 value: 2.30 μM (for AML cells)Target:in vitro: Quinacrine is a fluorescent probe for the conformational transitions of the cholinergic receptor protein in its membrane-bound state.[1] In the patient AML samples, Quinacrine showed activity in the low μM range with a mean IC50 of 2.30 μM, statistically significantly lower than that of normal PBMCs; 3.54 μM (P=0.0327; Student's t-test). Samples from patients with chronic lymphocytic, acute myeloid and lymphocytic leukemias as well as peripheral blood mononuclear cells (PBMC) were tested in response to 1266 compounds from the LOPAC1280 library. 25 compounds were defined as hits with activity in all leukemia subgroups (<50% cell survival compared with control) at 10 μM drug concentration. Only Quinacrine showed concurrent high activity in all leukemia subgroups and low activity in normal PBMCs and was, therefore, selected for further preclinical evaluation. Quinacrine also induced early inhibition of both DNA and protein synthesis. Quinacrine have repositioning potential for treatment of acute myeloid leukemia by targeting of ribosomal biogenesis.[2] |
| Name | Quinacrine Dihydrochloride Hydrate |
|---|---|
| Synonym | More Synonyms |
| Description | Quinacrine is a fluorescent probe for the conformational transitions of the cholinergic receptor protein. Quinacrine shows activity in the low μM range with a mean IC50 of 2.30 μM In the patient AML cells.IC50 value: 2.30 μM (for AML cells)Target:in vitro: Quinacrine is a fluorescent probe for the conformational transitions of the cholinergic receptor protein in its membrane-bound state.[1] In the patient AML samples, Quinacrine showed activity in the low μM range with a mean IC50 of 2.30 μM, statistically significantly lower than that of normal PBMCs; 3.54 μM (P=0.0327; Student's t-test). Samples from patients with chronic lymphocytic, acute myeloid and lymphocytic leukemias as well as peripheral blood mononuclear cells (PBMC) were tested in response to 1266 compounds from the LOPAC1280 library. 25 compounds were defined as hits with activity in all leukemia subgroups (<50% cell survival compared with control) at 10 μM drug concentration. Only Quinacrine showed concurrent high activity in all leukemia subgroups and low activity in normal PBMCs and was, therefore, selected for further preclinical evaluation. Quinacrine also induced early inhibition of both DNA and protein synthesis. Quinacrine have repositioning potential for treatment of acute myeloid leukemia by targeting of ribosomal biogenesis.[2] |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2962 (rough estimate) |
|---|---|
| Melting Point | ca. 248 - 250ºC (decomposes) |
| Molecular Formula | C23H32Cl3N3O |
| Molecular Weight | 472.88 |
| PSA | 37.39000 |
| LogP | 6.84740 |
| Index of Refraction | 1.6300 (estimate) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn |
| Risk Phrases | 22-36/37/38 |
| Safety Phrases | S36/S37/S39 |
| RIDADR | NONH for all modes of transport |
| RTECS | AR7875000 |
|
Chemical genetics reveals a complex functional ground state of neural stem cells.
Nat. Chem. Biol. 3(5) , 268-273, (2007) The identification of self-renewing and multipotent neural stem cells (NSCs) in the mammalian brain holds promise for the treatment of neurological diseases and has yielded new insight into brain canc... |
|
|
Genetic mapping of targets mediating differential chemical phenotypes in Plasmodium falciparum.
Nat. Chem. Biol. 5 , 765-71, (2009) Studies of gene function and molecular mechanisms in Plasmodium falciparum are hampered by difficulties in characterizing and measuring phenotypic differences between individual parasites. We screened... |
|
|
Real time visualization and characterization of platelet deposition under flow onto clinically relevant opaque surfaces.
J. Biomed. Mater. Res. A 103(4) , 1303-11, (2015) Although the thrombogenic nature of the surfaces of cardiovascular devices is an important aspect of blood biocompatibility, few studies have examined platelet deposition onto opaque materials used fo... |
| Atebrin hydrochloride |
| 866 R.P. |
| Chemiochin |
| Acrichine |
| Mepacrine dihydrochloride |
| MFCD00012659 |
| Chinacrin hydrochloride |
| Mecryl |
| Erion |
| Pentilen |
| N-(6-Chloro-2-methoxy-9-acridinyl)-N,N-diethyl-1,4-pentanediamine dihydrochloride dihydrate |
| QUINACRINE DIHYDROCHLORIDE |
| Palusan |
| 1,4-Pentanediamine, N-(6-chloro-2-methoxy-9-acridinyl)-N,N-diethyl-, hydrochloride, hydrate (1:2:2) |
| Crinodora |
| Akrichin |
| Italchin |
| Metochin |
| Metoquine |
| Palacrin |
| 69-05-6 {Anhydrous} |
| EINECS 200-700-8 |
| 1,4-Pentanediamine,N4-(6-chloro-2-methoxy-9-acridinyl)-N1,N1-diethyl-, hydrochloride (1:2) |
| Acriquine |
| Malaricida |
| Quinacrine (dihydrochloride) |
 CAS#:21332-86-5
CAS#:21332-86-5 CAS#:3548-09-2
CAS#:3548-09-2 CAS#:908844-46-2
CAS#:908844-46-2 CAS#:13161-87-0
CAS#:13161-87-0![6-chloro-9-[(4-diethylamino-1-methyl-butyl)amino]acridin-2-ol structure](https://www.chemsrc.com/caspic/443/854892-24-3.png) CAS#:854892-24-3
CAS#:854892-24-3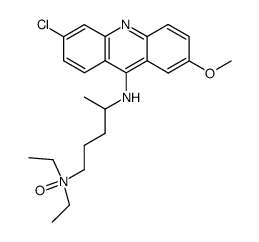 CAS#:83196-72-9
CAS#:83196-72-9 CAS#:3407-99-6
CAS#:3407-99-6
