Malvidin chloride
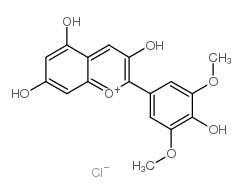
Malvidin chloride structure
|
Common Name | Malvidin chloride | ||
|---|---|---|---|---|
| CAS Number | 643-84-5 | Molecular Weight | 366.75000 | |
| Density | 1.183 g/mL at 25 °C(lit.) | Boiling Point | 66-67 °C(lit.) | |
| Molecular Formula | C17H15ClO7 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 30 °F | |
Use of Malvidin chlorideMalvidin (chloride) is a bioactive compound isolated from Oryza sativacv. Heugjinjubyeo. Malvidin shows cytotoxicity through the arrest of the G2/M phase of cell cycle and induction of apoptosis. Malvidin can be used for the research of cancer[1]. |
| Name | Malvidin chloride |
|---|---|
| Synonym | More Synonyms |
| Description | Malvidin (chloride) is a bioactive compound isolated from Oryza sativacv. Heugjinjubyeo. Malvidin shows cytotoxicity through the arrest of the G2/M phase of cell cycle and induction of apoptosis. Malvidin can be used for the research of cancer[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Malvidin (0~80 μg/mL; 96 hours; U937 cells) shows cytotoxic activity in a dose-dependent pattern[1]. The IC50 value of Malvidin for U937 cells is 40 μg/mL. Malvidin (U937 cells) shows arrest at the G2/M phase of the cell cycle and increases sub G1 hypo-diploid population[1]. Cell Viability Assay[1] Cell Line: U937 cells Concentration: 0~80 μg/mL Incubation Time: 96 hours Result: Showed cytotoxic activity in a dose-dependent pattern. |
| References |
| Density | 1.183 g/mL at 25 °C(lit.) |
|---|---|
| Boiling Point | 66-67 °C(lit.) |
| Molecular Formula | C17H15ClO7 |
| Molecular Weight | 366.75000 |
| Flash Point | 30 °F |
| Exact Mass | 366.05100 |
| PSA | 112.52000 |
| LogP | 0.22450 |
| Index of Refraction | n20/D 1.335(lit.) |
| Storage condition | ?20°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | LK9900000 |
| HS Code | 2909499000 |
|
~% 
Malvidin chloride CAS#:643-84-5 |
| Literature: Tetrahedron Letters, , vol. 23, # 38 p. 3963 - 3964 |
|
~% 
Malvidin chloride CAS#:643-84-5 |
| Literature: Tetrahedron Letters, , vol. 23, # 38 p. 3963 - 3964 |
|
~% 
Malvidin chloride CAS#:643-84-5 |
| Literature: Tetrahedron Letters, , vol. 23, # 38 p. 3963 - 3964 |
|
~% 
Malvidin chloride CAS#:643-84-5 |
| Literature: Phytochemistry, , vol. 70, # 5 p. 672 - 674 |
|
~% 
Malvidin chloride CAS#:643-84-5 |
| Literature: Phytochemistry, , vol. 70, # 5 p. 672 - 674 |
|
~% 
Malvidin chloride CAS#:643-84-5 |
| Literature: Journal of the Chemical Society, , p. 1551 |
| HS Code | 2909499000 |
|---|---|
| Summary | 2909499000. ether-alcohols and their halogenated, sulphonated, nitrated or nitrosated derivatives. VAT:17.0%. Tax rebate rate:9.0%. . MFN tariff:5.5%. General tariff:30.0% |
|
A nicotinic receptor-mediated anti-inflammatory effect of the flavonoid rhamnetin in BV2 microglia.
Fitoterapia 98 , 11-21, (2014) The alpha7 nicotinic acetylcholine receptor (nAChR) is a potential target in neuroinflammation. Screening a plant extract library identified Solidago nemoralis as containing methyl-quercetin derivativ... |
|
|
How subtle is the "terroir" effect? Chemistry-related signatures of two "climats de Bourgogne".
PLoS ONE 9(5) , e97615, (2014) The chemical composition of grape berries is influenced by various environmental conditions often considered to be representative of a "terroir". If grapes from a given terroir are assumed to reflect ... |
|
|
Direct and indirect inactivation of tumor cell protective catalase by salicylic acid and anthocyanidins reactivates intercellular ROS signaling and allows for synergistic effects.
Carcinogenesis 36(3) , 400-11, (2015) Salicylic acid and anthocyanidins are known as plant-derived antioxidants, but also can provoke paradoxically seeming prooxidant effects in vitro. These prooxidant effects are connected to the potenti... |
| MFCD00017585 |
| 2-(4-hydroxy-3,5-dimethoxyphenyl)chromenylium-3,5,7-triol,chloride |
| EINECS 211-403-8 |


![malvidin 3-O-[6-O-(3-hydroxy-3-methylglutaroyl)-β-D-glucopyranoside] structure](https://www.chemsrc.com/caspic/385/1158964-28-3.png)

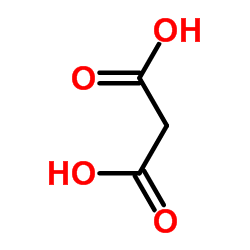
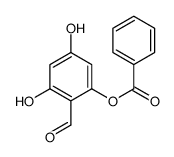
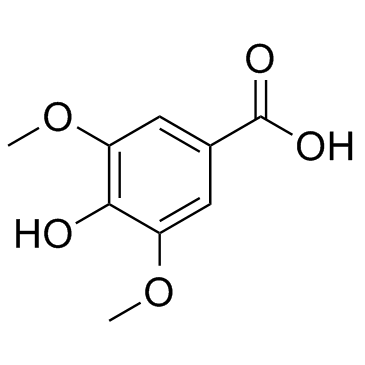 CAS#:530-57-4
CAS#:530-57-4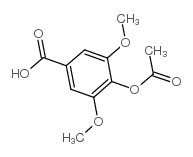 CAS#:6318-20-3
CAS#:6318-20-3
