4-Bromoindole
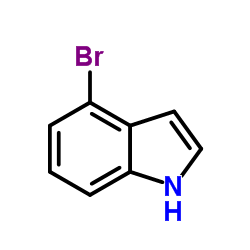
4-Bromoindole structure
|
Common Name | 4-Bromoindole | ||
|---|---|---|---|---|
| CAS Number | 52488-36-5 | Molecular Weight | 196.044 | |
| Density | 1.7±0.1 g/cm3 | Boiling Point | 316.9±15.0 °C at 760 mmHg | |
| Molecular Formula | C8H6BrN | Melting Point | 17 °C | |
| MSDS | Chinese USA | Flash Point | 145.5±20.4 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | 4-bromo-1H-indole |
|---|---|
| Synonym | More Synonyms |
| Density | 1.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 316.9±15.0 °C at 760 mmHg |
| Melting Point | 17 °C |
| Molecular Formula | C8H6BrN |
| Molecular Weight | 196.044 |
| Flash Point | 145.5±20.4 °C |
| Exact Mass | 194.968353 |
| PSA | 15.79000 |
| LogP | 2.91 |
| Vapour Pressure | 0.0±0.7 mmHg at 25°C |
| Index of Refraction | 1.712 |
| Storage condition | 0-6°C |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2933990090 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
A concise and scalable strategy for the total synthesis of dictyodendrin B based on sequential C-H functionalization.
Angew. Chem. Int. Ed. Engl. 54(18) , 5451-5, (2015) A sequential CH functionalization strategy for the synthesis of the marine alkaloid dictyodendrin B is reported. Our synthesis begins from commercially available 4-bromoindole and involves six direct... |
|
|
Applications of vinylogous Mannich reactions. Total syntheses of the Ergot alkaloids rugulovasines A and B and setoclavine.
J. Am. Chem. Soc. 123(25) , 5918-24, (2001) Concise syntheses of the Ergot alkaloids rugulovasine A (3a), rugulovasine B (3b), and setoclavine (2) have been completed by strategies that feature inter- and intramolecular vinylogous Mannich react... |
|
|
Optically active total synthesis of clavicipitic acid. Yokoyama Y, et al.
J. Org. Chem. 60(6) , 1486-1487, (1995)
|
| 5-Br-indole |
| 4-bromo-indole |
| 5-Bromo-1H-indole |
| 5-bromo-indole |
| 4-bromindole |
| MFCD00671502 |
| 5-BI |
| 1H-INDOLE,4-BROMO |
| 4-Bromo-1H-indole |
| 5-broMo-2H-indole |
| 1H-Indole, 4-bromo- |
| 1H-Indole, 5-bromo- |
| 5-bromonindole |
| 5-Bromide indole |
| 5-indolyl bromide |
| 4-Brom-indol |
| 5-Bromindole |
| 4-Bromoindole |
| 5-Bromoindole |
| Bromoindole |
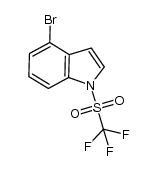 CAS#:1180007-97-9
CAS#:1180007-97-9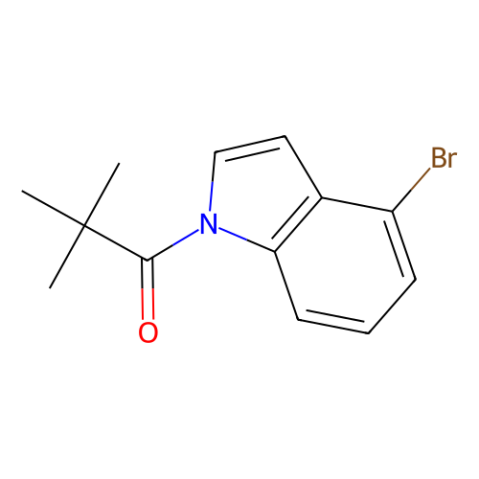 CAS#:1196981-04-0
CAS#:1196981-04-0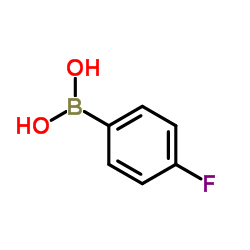 CAS#:1765-93-1
CAS#:1765-93-1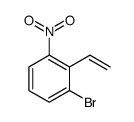 CAS#:90481-68-8
CAS#:90481-68-8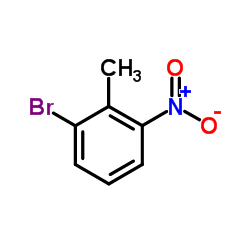 CAS#:55289-35-5
CAS#:55289-35-5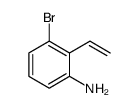 CAS#:90481-69-9
CAS#:90481-69-9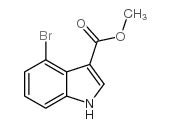 CAS#:101909-43-7
CAS#:101909-43-7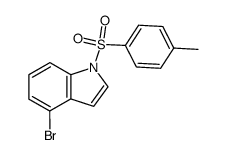 CAS#:90481-72-4
CAS#:90481-72-4 CAS#:388116-27-6
CAS#:388116-27-6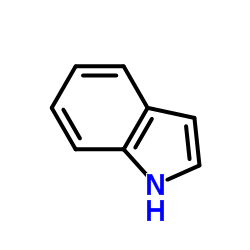 CAS#:120-72-9
CAS#:120-72-9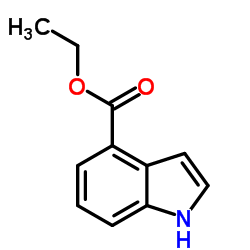 CAS#:50614-84-1
CAS#:50614-84-1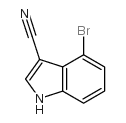 CAS#:903131-13-5
CAS#:903131-13-5![4-Bromo-3-[4-(chloromethyl)-3-fluorobenzoyl]-N,N-dimethyl-1H-indo le-1-carboxamide structure](https://www.chemsrc.com/caspic/312/200418-19-5.png) CAS#:200418-19-5
CAS#:200418-19-5![3-[4-(aminomethyl)-3-fluorobenzoyl]-4-bromo-N,N-dimethylindole-1-carboxamide structure](https://www.chemsrc.com/caspic/419/200418-21-9.png) CAS#:200418-21-9
CAS#:200418-21-9![4-bromo-3-[3-fluoro-4-[[(3-nitropyridin-4-yl)amino]methyl]benzoyl]-N,N-dimethylindole-1-carboxamide structure](https://www.chemsrc.com/caspic/371/200418-22-0.png) CAS#:200418-22-0
CAS#:200418-22-0 CAS#:73-22-3
CAS#:73-22-3 CAS#:86626-38-2
CAS#:86626-38-2 CAS#:98600-34-1
CAS#:98600-34-1
