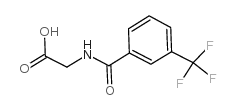
1341224-83-6
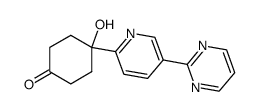
| Name | (S)-N-[2-(3-{trans-4-hydroxy-4-[5-(pyrimidin-2-yl)pyridin-2-yl]cyclohexylamino}pyrrolidin-1-yl)-2-oxoethyl]-3-(trifluoromethyl)benzamide |
|---|---|
| Synonyms |
incb8761
PF-4136309 |
| Description | PF-4136309 is a potent, selective, and orally bioavailable CCR2 antagonist, with IC50 of 5.2 nM, 17 nM and 13 nM for human, mouse and rat CCR2. |
|---|---|
| Related Catalog | |
| Target |
Human CCR2:5.2 nM (IC50) Mouse CCR2:13 nM (IC50) Rat CCR2:17 nM (IC50) |
| In Vitro | PF-4136309 is potent in human chemotaxis activity (IC50=3.9 nM) and in the whole blood assay (IC50=19 nM), with IC50 of 16 and 2.8 nM in mouse and rat chemotaxis assays. PF-4136309 is potent in inhibiting CCR2 mediated signaling events such as intracellular calcium mobilization and ERK (extracellular signal-regulated kinase) phosphorylation with IC50 values of 3.3 and 0.5 nM, respectively. In hERG patch clamp assay, PF-4136309 inhibits hERG potassium current with an IC50 of 20 μM. PF-4136309 is not a cytochrome P450 (CYP) inhibitor, with IC50 values of >30 μM against five major CYP isozymes CYP1A2, CYP2C9, CYP2C19, CYP2D6, and CYP3A4. Moreover, PF-4136309 is not a CYP inducer at concentrations up to 30 μM[1]. |
| In Vivo | PF-4136309 (2 mg/kg) exhibits a moderate half-life in both species after iv administration (2.5 and 2.4 h). When administered orally, PF-4136309 (10 mg/kg) is absorbed rapidly, with peak concentration time (Tmax) at 1.2 h for rats and 0.25 h for dogs. A similar half-life is observed in both species between iv dosing and po dosing. PF-4136309 is well absorbed, with an oral bioavailability of 78% in both species[1]. |
| References |
| Molecular Formula | C29H31F3N6O3 |
|---|---|
| Molecular Weight | 568.59000 |
| Exact Mass | 568.24100 |
| PSA | 120.34000 |
| LogP | 4.02780 |
| Storage condition | 2-8℃ |
|
~65% 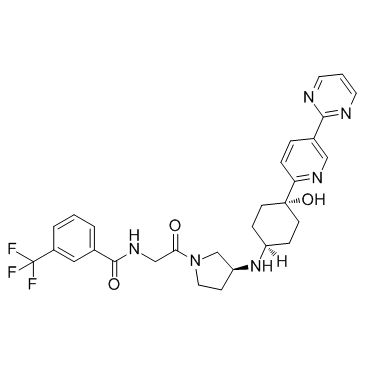
1341224-83-6 |
| Literature: Xue, Chu-Biao; Wang, Anlai; Han, Qi; Zhang, Yingxin; Cao, Ganfeng; Feng, Hao; Huang, Taisheng; Zheng, Changsheng; Xia, Michael; Zhang, Ke; Kong, Lingquan; Glenn, Joseph; Anand, Rajan; Meloni, David; Robinson; Shao, Lixin; Storace, Lou; Li, Mei; Hughes, Robert O.; Devraj, Rajesh; Morton, Philip A.; Rogier, D. Joseph; Covington, Maryanne; Scherle, Peggy; Diamond, Sharon; Emm, Tom; Yeleswaram, Swamy; Contel, Nancy; Vaddi, Kris; Newton, Robert; Hollis, Greg; Metcalf, Brian ACS Medicinal Chemistry Letters, 2011 , vol. 2, # 12 p. 913 - 918 |
|
~% 
1341224-83-6 |
| Literature: ACS Medicinal Chemistry Letters, , vol. 2, # 12 p. 913 - 918 |
|
~% 
1341224-83-6 |
| Literature: ACS Medicinal Chemistry Letters, , vol. 2, # 12 p. 913 - 918 |
|
~% 
1341224-83-6 |
| Literature: ACS Medicinal Chemistry Letters, , vol. 2, # 12 p. 913 - 918 |
|
~% 
1341224-83-6 |
| Literature: ACS Medicinal Chemistry Letters, , vol. 2, # 12 p. 913 - 918 |
|
~% 
1341224-83-6 |
| Literature: ACS Medicinal Chemistry Letters, , vol. 2, # 12 p. 913 - 918 |
| Precursor 6 | |
|---|---|
| DownStream 0 | |