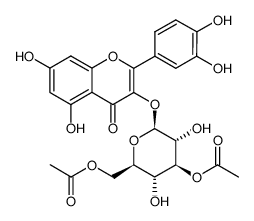
482-35-9
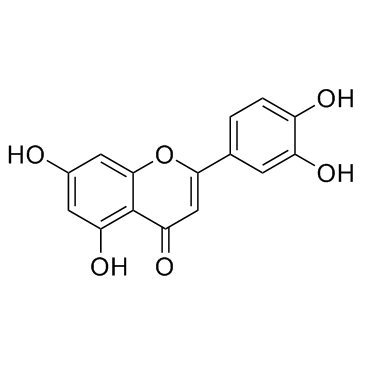
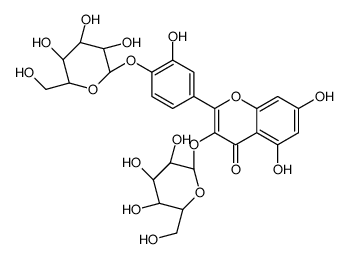
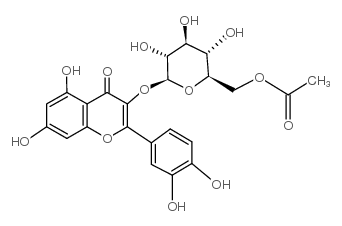
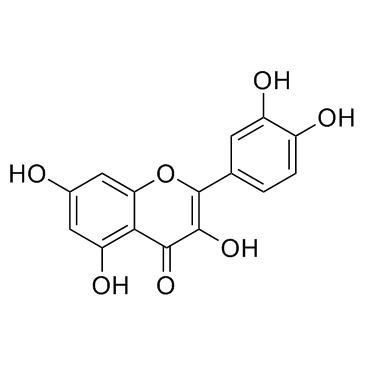
| Name | 3,3′,4′,5,7-Pentahydroxyflavone 3-β-glucoside, Isoquercitrin, Quercetin 3-β-D-glucoside |
|---|---|
| Synonyms |
Quercetin 3-O-β-glucoside
trifoliin quercetin 3-O-β-D-glucopyranoside Quercetin 3β-O-glucoside Quercetin 3-β-glucoside 2-(3,4-Dihydroxyphenyl)-5,7-dihydroxy-4-oxo-4H-chromen-3-yl β-D-glucopyranoside 2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-3-[(3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxychromen-4-one Quercetin 3-β-D-glucoside 3-O-β-D-Glucopyranosylquercetin Quercetin glucoside Quercetin-3-O-β-D-glucoside Quercetin-3-O-β-glucopyranoside Isoquercetrin Quercetin 3-O-glucoside Quercetin 3-D-glucoside Isoquercitrin isoquercetin quercetin 3-O-β-D-glucoside Hirsutrin isotrifoliin 4H-1-Benzopyran-4-one, 2-(3,4-dihydroxyphenyl)-3-(β-D-glucopyranosyloxy)-5,7-dihydroxy- Quercetin 3-glucoside Quercetin-3-β-glucopyranoside Quercetin-3-O-glucoside Quercetin 3-β-O-glucoside Quercetin 3-O-glucopyranoside |
| Description | Quercetin-3-glucoside is a naturally occurring polyphenol that has antioxidant, anti-proliferative, and anti-inflammatory properties.Quercetin-3-glucoside alleviates ethanol-induced hepatotoxicity, oxidative stress, and inflammatory responses via the Nrf2/ARE antioxidant signaling pathway[1].Quercetin-3-glucoside regulates the expression of nitric oxide synthase 2 (NO2) via modulating the nuclear factor-κB (NF-κB) transcription regulation system. Quercetin-3-glucoside has high bioavailability and low toxicity, is a promising candidate agent to prevent birth defects in diabetic pregnancies[2]. |
|---|---|
| Related Catalog | |
| In Vitro | Quercetin-3-glucoside (5-20 μM; 24 hours) substantially reduces ethanol-induced cytotoxicity , protects hepatic cells against ethanol‐stimulated liver injury[1].Quercetin-3-glucoside (10 μM; pre-treat 1 hour) dramatically downregulates the levels of ethanol-induced iNOS protein expression in HepG2 cells[1]. Cell Viability Assay[1] Cell Line: HepG2 cells Concentration: 5 μM, 10 μM, 20 μM Incubation Time: 24 hours Result: Caused significantly enhanced cell viability as positive controls. Western Blot Analysis[1] Cell Line: HepG2 cells Concentration: 10 μM Incubation Time: 1 hour Result: Decreased ethanol‐ induced iNOS protein expression. |
| References |
| Density | 1.9±0.1 g/cm3 |
|---|---|
| Boiling Point | 872.6±65.0 °C at 760 mmHg |
| Molecular Formula | C21H20O12 |
| Molecular Weight | 464.376 |
| Flash Point | 307.5±27.8 °C |
| Exact Mass | 464.095490 |
| PSA | 210.51000 |
| LogP | 1.75 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.803 |
| Storage condition | −20°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi: Irritant; |
| Safety Phrases | 24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | LK8960000 |
| Precursor 5 | |
|---|---|
| DownStream 10 | |

![2-(2,2-diphenylbenzo[d][1,3]dioxol-5-yl)-3,5,7-trihydroxy-4H-chroMen-4-one structure](https://www.chemsrc.com/caspic/359/357194-03-7.png)