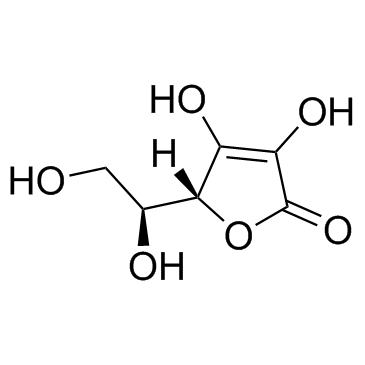
129499-78-1
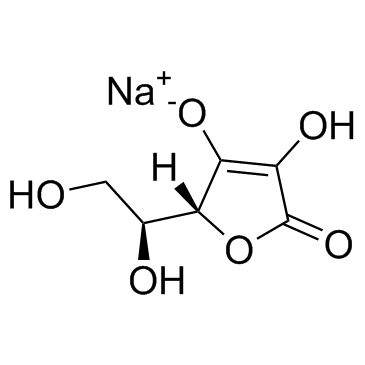
| Name | Ascorbyl Glucoside |
|---|---|
| Synonyms |
(2R)-2-[(1S)-1,2-dihydroxyethyl]-3-hydroxy-4-[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-2H-furan-5-one
(5R)-5-[(1S)-1,2-Dihydroxyethyl]-4-hydroxy-3-{[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl]oxy}-2(5H)-furanone 2-O-α-D-Glucopyranosyl-L-ascorbic Acid 2-O-alpha-D-Glucopyranosyl-L-ascorbic acid |
| Description | 2-O-α-D-Glucopyranosyl-L-ascorbic Acid is a glucoside derivative of ascorbic acid, shows anti-cancer activity after enzymatic hydrolysis to ascorbic acid[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.83±0.1 g/cm3 |
|---|---|
| Boiling Point | 785.6±60.0 °C at 760 mmHg |
| Melting Point | 158-163℃ |
| Molecular Formula | C12H18O11 |
| Molecular Weight | 338.265 |
| Flash Point | 296.5±26.4 °C |
| Exact Mass | 338.084900 |
| PSA | 186.37000 |
| LogP | -2.57 |
| Appearance | Powder | White to Off-white |
| Vapour Pressure | 0.0±6.2 mmHg at 25°C |
| Index of Refraction | 1.670 |
| Storage condition | 2-8°C |
| Water Solubility | Freely soluble (879 g/L) (25 ºC) |
| RIDADR | NONH for all modes of transport |
|---|---|
| HS Code | 2936270090 |
|
~0% 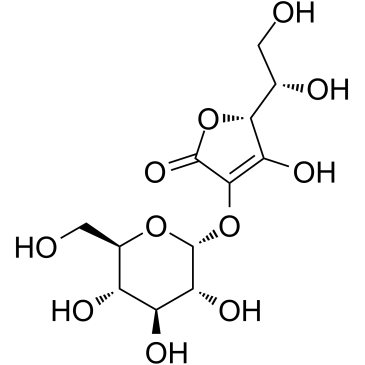
129499-78-1 |
| Literature: Journal of Molecular Catalysis B: Enzymatic, , vol. 92, p. 19 - 23 |
|
~0% 
129499-78-1 |
| Literature: Journal of Molecular Catalysis B: Enzymatic, , vol. 92, p. 19 - 23 |
|
~0% 
129499-78-1 |
| Literature: Journal of Molecular Catalysis B: Enzymatic, , vol. 92, p. 19 - 23 |
|
~% 
129499-78-1 |
| Literature: Journal of Molecular Catalysis B: Enzymatic, , vol. 68, # 3-4 p. 223 - 229 |
|
~% 
129499-78-1 |
| Literature: Agricultural and Biological Chemistry, , vol. 54, # 7 p. 1697 - 1703 |
|
~% 
129499-78-1 |
| Literature: Agricultural and Biological Chemistry, , vol. 55, # 7 p. 1751 - 1756 |
|
~14% 
129499-78-1 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 38, # 11 p. 3020 - 3023 |
| Precursor 6 | |
|---|---|
| DownStream 0 | |
| HS Code | 2936270090 |
|---|