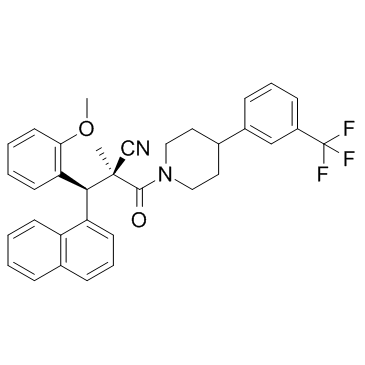
796854-35-8
| Name | WAY-204688 |
|---|---|
| Synonyms |
1-Piperidinepropanenitrile, α-[(S)-(2-methoxyphenyl)-1-naphthalenylmethyl]-α-methyl-β-oxo-4-[3-(trifluoromethyl)phenyl]-, (αS)-
(2S,3S)-3-(2-Methoxyphenyl)-2-methyl-3-(1-naphthyl)-2-({4-[3-(trifluoromethyl)phenyl]-1-piperidinyl}carbonyl)propanenitrile (2S,3S)-3-(2-methoxyphenyl)-2-methyl-3-(naphthalen-1-yl)-2-({4-[3-(trifluoromethyl)phenyl]piperidin-1-yl}carbonyl)propanenitrile |
| Description | WAY-204688 is an estrogen receptor (ER-α) selective, orally active inhibitor of NF-κB transcriptional activity with an IC50 of 122 ± 30 nM for NF-κB-luciferase (NF-κB-luc) in HAECT-1 cells. |
|---|---|
| Related Catalog | |
| Target |
NF-κB-luc:122 nM (IC50, in HAECT-1 cell) NF-κB |
| In Vitro | WAY-204688 is ER-dependenrt (activity seen only when hER is coexpressed with NF-κB-luciferase in human aortic endothelial cell lines (HAECT-1) cells). The interaction of WAY-204688 with ERα and ERβ is examined in vitro. WAY-204688 displaces [3H]E2 from the ERα ligand binding domain protein (LBD) with IC50=2.43 μM and from the ERβ ligand binding domain protein (LBD) with IC50=1.5 μM[1]. |
| In Vivo | WAY-204688 (5 mg/kg per day, po daily for 5 weeks) is evaluated in vivo for the ability to inhibit four proinflammatory genes (MHC, invariant chain (MHI), VCAM-1, RANTES, and TNF-α). The effect of WAY-204688 on induction of the gene products and on uterine wet weight is compared to that of 17α-ethinyl 17β-estradiol (EE at 10 μg/kg per day) in the same paradigm. Further characterization of WAY-204688 is carried out in several preclinical models of inflammatory disease. In the Lewis rat adjuvant-induced arthritis model (AIA), WAY-204688 is active at a dose of 0.3 mg/kg per day, po[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 708.7±60.0 °C at 760 mmHg |
| Molecular Formula | C34H31F3N2O2 |
| Molecular Weight | 556.617 |
| Flash Point | 382.4±32.9 °C |
| Exact Mass | 556.233765 |
| LogP | 7.99 |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.589 |
| Storage condition | 2-8℃ |