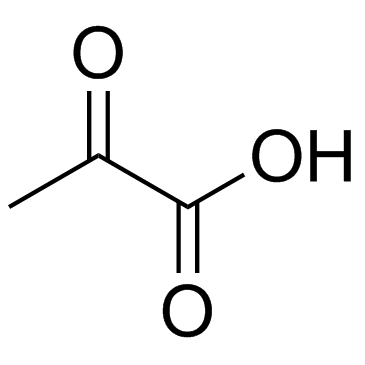
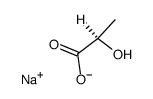
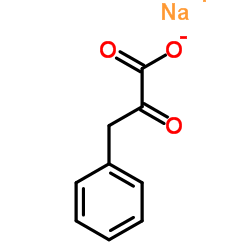
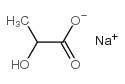
113-24-6
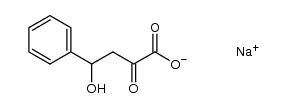
| Name | sodium pyruvate |
|---|---|
| Synonyms |
PYRUVATE
PYR NA Pyruvic acid,sodium salt sodium 2-oxopropanoate Sodium pyroracemate p.A.,100g Sodium pyruate SodiuM Pyruvate Pyruvic acid sodium salt pyruvic acid sodium 2-oxopropanoic acid sodium salt sodium salt of pyruvic acid EINECS 204-024-4 SodiuM pyrovaceMate BioChemica,100g MFCD00002586 |
| Description | Sodium 2-oxopropanoate (Sodium pyruvate), a three-carbon metabolite of Glucose, is a compound produced in the glycolytic pathway. Sodium 2-oxopropanoate is a free radical scavenger that can scavenge ROS[1][2]. |
|---|---|
| Related Catalog | |
| Target |
Microbial Metabolite |
| In Vitro | In the process of scavenging hydrogen peroxide, Sodium 2-oxopropanoate (Sodium pyruvate) is decarboxylated to lactate and hence diverted away from aerobic metabolism and further ROS generation. Under conditions in which pyruvate stimulated JNK1 activity, both mitochondrial and cytosolic ROS levels rose. An increase in JNK1 activity in a variety of different cell types over a range of pyruvate concentrations are observed[1]. Sodium 2-oxopropanoate (Sodium pyruvate) is an effective scavenger of H2O2 as well as of O2▪-, thereby protecting the lens against oxidative stress and consequent cataract formation, under in vitro as well as in vivo conditions, Sodium 2-oxopropanoate has also been shown to protect the lens proteins against glycation by competitively inhibiting the initial reaction between the sugar carbonyl and the protein -NH2[2]. |
| In Vivo | Neonatal rats are given a bolus of Sodium 2-oxopropanoate (Sodium pyruvate; 0.1-10 g/kg), and 1 h later, levels of JNK1 activity were determined in liver extracts. Sodium 2-oxopropanoate (Sodium pyruvate) addition resulted in a significant increase in JNK1 activity. Analysis of serum levels of Sodium 2-oxopropanoate in neonatal rats revealed that baseline levels of pyruvate are 0.30 mM. At the time of sacrifice, following the largest dose of Sodium 2-oxopropanoate, serum levels rose approximately sixfold to 1.84 mM[1]. |
| References |
| Density | 1.267g/cm3 |
|---|---|
| Boiling Point | 165ºC at 760 mmHg |
| Melting Point | >300 °C(lit.) |
| Molecular Formula | C3H3NaO3 |
| Molecular Weight | 110.04 |
| Flash Point | 54.3ºC |
| Exact Mass | 109.997986 |
| PSA | 57.20000 |
| Vapour Pressure | 0.968mmHg at 25°C |
| Index of Refraction | 1.426-1.43 |
| Storage condition | 2-8°C |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Water Solubility | H2O: 100 mg/mL |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S37/39-S26-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 29183000 |
|
~93% 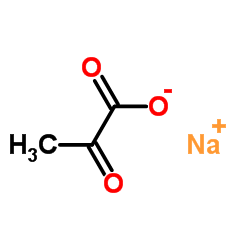
113-24-6 |
| Literature: GE HEALTHCARE AS Patent: WO2008/48106 A1, 2008 ; Location in patent: Page/Page column 18 ; |
|
~94% 
113-24-6 |
| Literature: SKW Trostberg Aktiengesellschaft Patent: US6232497 B1, 2001 ; |
|
~% 
113-24-6 |
| Literature: Process Biochemistry, , vol. 48, # 1 p. 109 - 117 |
|
~%
Detail
|
| Literature: Journal of Organic Chemistry, , vol. 59, # 11 p. 3227 - 3229 |
|
~% 
113-24-6 |
| Literature: Angewandte Chemie - International Edition, , vol. 48, # 5 p. 922 - 925 |
|
~% 
113-24-6 |
| Literature: Journal of the Chemical Society, Perkin Transactions 2: Physical Organic Chemistry (1972-1999), , p. 1919 - 1924 |
|
~% 
113-24-6 |
| Literature: Journal of the Chemical Society, Perkin Transactions 2: Physical Organic Chemistry (1972-1999), , # 2 p. 263 - 267 |
|
~% 
113-24-6 |
| Literature: Bulletin de la Societe Chimique de France, , vol. 132, # 10 p. 1039 - 1046 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2918300090 |
|---|---|
| Summary | 2918300090 other carboxylic acids with aldehyde or ketone function but without other oxygen function, their anhydrides, halides, peroxides, peroxyacids and their derivatives。Supervision conditions:None。VAT:17.0%。Tax rebate rate:9.0%。MFN tariff:6.5%。General tariff:30.0% |