Iron dextran
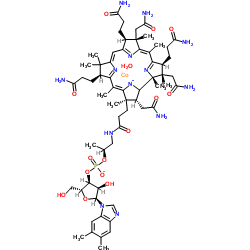
Iron dextran structure
|
Common Name | Iron dextran | ||
|---|---|---|---|---|
| CAS Number | 9004-66-4 | Molecular Weight | 1347.364 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | H2O4S.Fe | Melting Point | N/A | |
| MSDS | USA | Flash Point | N/A | |
| Symbol |


GHS07, GHS08 |
Signal Word | Danger | |
Use of Iron dextranIron dextran (Fe dextran) can be used in the study of iron-deficiency anemia in animals[1][2]. |
| Name | Iron-dextran |
|---|---|
| Synonym | More Synonyms |
| Description | Iron dextran (Fe dextran) can be used in the study of iron-deficiency anemia in animals[1][2]. |
|---|---|
| Related Catalog | |
| In Vivo | Iron dextran (FeDex, IP, 4 weeks, 300 μL of 10 mg) treatment does not increase NSR iron but causes iron-trapping in the tetinal vasculature[1]. Four weeks of IP FeDex injection in 2-month-old WT mice leads to iron accumulation in the RPE and rVECs, but did not increase NSR iron or induce photoreceptor degeneration[1]. Animal Model: Wild-type (WT) and RS-HepcKO mice[1]. Dosage: IP. Administration: 300 μL of 10 mg daily for 5 days each week for 2 or 4 weeks. Result: Markedly elevated iron levels in 2 and 4 weeks of FeDex-injection groups, by 365.4-fold and 405.4-fold, respectively, compared with PBS controls. |
| References |
| Molecular Formula | H2O4S.Fe |
|---|---|
| Molecular Weight | 1347.364 |
| Exact Mass | 1346.576050 |
| Appearance of Characters | solution |
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H317-H350 |
| Precautionary Statements | P201-P280-P308 + P313 |
| Hazard Codes | Xn |
| Risk Phrases | R40;R42/43 |
| Safety Phrases | S23;S26;S45;S36/S37/S39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | - |
| RTECS | NI2200000 |
|
Deferasirox shows in vitro and in vivo antileukemic effects on murine leukemic cell lines regardless of iron status
Exp. Hematol. 41(6) , 539-46, (2013) Numerous studies have shown the antiproliferative effect of iron chelating agents (ICAs), which have been used traditionally in patients with secondary iron overload (SIO). Because the in vivo model f... |
|
|
[Importance of the different i.v. iron generations for everyday medical practice].
MMW Fortschr. Med. 155 Suppl 1 , 18-24, (2013) Iron deficiency and anaemia occur in particular in women or as comorbid conditions to a varietyof chronic diseases. Besides oral preparations, parenteral iron therapies are also available for the trea... |
|
|
Use of intravenous iron supplementation in chronic kidney disease: an update.
Iran. J. Kidney Dis. 7(1) , 9-22, (2013) Iron deficiency is an important clinical concern in chronic kidney disease (CKD), giving rise to iron-deficiency anemia and impaired cellular function. Oral supplementation, in particular with ferrous... |
| MFCD00081553 |
| Ferric hydroxide dextran complex |