Sulprostone
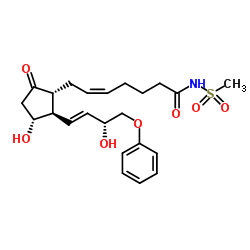
Sulprostone structure
|
Common Name | Sulprostone | ||
|---|---|---|---|---|
| CAS Number | 60325-46-4 | Molecular Weight | 465.560 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C23H31NO7S | Melting Point | 78.5-80ºC | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |


GHS07, GHS08 |
Signal Word | Danger | |
Use of SulprostoneSulprostone (SHB 286), a prostaglandin E2 (PGE2) analogue, is a potent and selective EP3 receptor agonist. Sulprostone has potential antiulcer and nonsteroidal abortifacient effects used for the research of pregnancy termination and hemorrhages during delivery[1][2][3]. |
| Name | (Z)-7-[(1R,2R,3R)-3-hydroxy-2-[(E,3R)-3-hydroxy-4-phenoxybut-1-enyl]-5-oxocyclopentyl]-N-methylsulfonylhept-5-enamide |
|---|---|
| Synonym | More Synonyms |
| Description | Sulprostone (SHB 286), a prostaglandin E2 (PGE2) analogue, is a potent and selective EP3 receptor agonist. Sulprostone has potential antiulcer and nonsteroidal abortifacient effects used for the research of pregnancy termination and hemorrhages during delivery[1][2][3]. |
|---|---|
| Related Catalog | |
| Target |
EP3 Receptor |
| In Vitro | Sulprostone (SHB 286) has Ki values of 21 nM and 0.6 nM for EP1 and EP3 in cultured Chinese hamster ovary cells[1]. Sulprostone (1, 1.5 or 2 mg/mL) does not significantly modify the viability and the purity of Dendritic cells (DCs). Sulprostone impairs spontaneous and directed DC migration independently from its concentration. Sulprostone reduces the expression of CCR7 and impairs migration of DCs[2]. |
| In Vivo | Sulprostone (0.5 mg/kg; IV) has a T1/2 of 0.451 hours, a CL of 56 mL/min•kg, a Vss of 0.583 L/kg and an AUC of 149 ng•h/mL[3]. Animal Model: Male cynomolgus monkey[3] Dosage: 0.5 mg/kg (Pharmacokinetic Analysis) Administration: IV Result: Had a T1/2 of 0.451 hours, a CL of 56 mL/min•kg, a Vss of 0.583 L/kg and an AUC of 149 ng•h/mL. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Melting Point | 78.5-80ºC |
| Molecular Formula | C23H31NO7S |
| Molecular Weight | 465.560 |
| Exact Mass | 465.182129 |
| PSA | 138.38000 |
| LogP | 0.26 |
| Index of Refraction | 1.589 |
| Storage condition | −20°C |
| Water Solubility | DMSO: >5 mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H315-H319-H335-H360 |
| Precautionary Statements | P201-P261-P305 + P351 + P338-P308 + P313 |
| Personal Protective Equipment | Eyeshields;full-face respirator (US);Gloves;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | T:Toxic |
| Risk Phrases | R60;R36/37/38 |
| Safety Phrases | 53-22-36/37-45 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | MJ8810000 |
| Precursor 10 | |
|---|---|
| DownStream 0 | |
|
Quantitative phosphoproteomic analysis of signaling downstream of the prostaglandin e2/g-protein coupled receptor in human synovial fibroblasts: potential antifibrotic networks.
J. Proteome Res. 13(11) , 5262-80, (2014) The Prostaglandin E2 (PGE2) signaling mechanism within fibroblasts is of growing interest as it has been shown to prevent numerous fibrotic features of fibroblast activation with limited evidence of d... |
|
|
Angiogenesis in the primate ovulatory follicle is stimulated by luteinizing hormone via prostaglandin E2.
Biol. Reprod. 92(1) , 15, (2015) Rapid angiogenesis occurs as the ovulatory follicle is transformed into the corpus luteum. To determine if luteinizing hormone (LH)-stimulated prostaglandin E2 (PGE2) regulates angiogenesis in the ovu... |
|
|
PGE2 reduces MMP-14 and increases plasminogen activator inhibitor-1 in cardiac fibroblasts.
Prostaglandins Other Lipid Mediat. 113-115 , 62-8, (2014) Prostaglandin E2 (PGE2) is elevated during cardiac injury and we have previously shown that mice lacking the PGE2 EP4 receptor display dilated cardiomyopathy (DCM) with increased expression of the mem... |
| Sulprostonum |
| N-Acetyl-N-acetoxy-4-chlorobenzenesulfonamide |
| Nalador |
| [1R-[1a(Z),2b(1E,3R*),3a]]-7-[3-Hydroxy-2-(3-hydroxy-4-phenoxy-1-butenyl)-5-oxocyclopentyl]-N-(methylsulfonyl)-5-heptenamide |
| Sulprostone |
| SHB-286 |
| Sulprostona |
| (5Z)-7-{(1R,2R,3R)-3-Hydroxy-2-[(1E,3R)-3-hydroxy-4-phenoxybut-1-en-1-yl]-5-oxocyclopentyl}-N-(methylsulfonyl)hept-5-enamide |
| (5Z)-7-{(1R,2R,3R)-3-Hydroxy-2-[(1E,3R)-3-hydroxy-4-phenoxy-1-buten-1-yl]-5-oxocyclopentyl}-N-(methylsulfonyl)-5-heptenamide |
| 5-Heptenamide, 7-[(1R,2R,3R)-3-hydroxy-2-[(1E,3R)-3-hydroxy-4-phenoxy-1-buten-1-yl]-5-oxocyclopentyl]-N-(methylsulfonyl)-, (5Z)- |
| SHB 286 |
| EINECS 262-173-0 |
 CAS#:64282-82-2
CAS#:64282-82-2![7α-hydroxy-6β-(3'-hydroxy-4'-phenoxy-1'E-butenyl)-2-oxabicyclo[3.3.0]octan-3-one Structure](https://www.chemsrc.com/caspic/359/51638-93-8.png) CAS#:51638-93-8
CAS#:51638-93-8![(3aR,4R,5R,6aS)-4-((3R,E)-4-phenoxy-3-((tetrahydro-2H-pyran-2-yl)oxy)but-1-en-1-yl)-5-((tetrahydro-2H-pyran-2-yl)oxy)hexahydro-2H-cyclopenta[b]furan-2-ol Structure](https://www.chemsrc.com/caspic/169/286840-19-5.png) CAS#:286840-19-5
CAS#:286840-19-5![(3aR,4R,5R,6aS)-Hexahydro-4-[(E)-(3R)-4-phenoxy-3-[(tetrahydro-2H-pyran-2-yl)oxy]-1-butenyl]-5-[(tetrahydro-2H-pyran-2-yl)oxy]-2H-cyclopenta[b]furan-2-one Structure](https://www.chemsrc.com/caspic/353/54347-99-8.png) CAS#:54347-99-8
CAS#:54347-99-8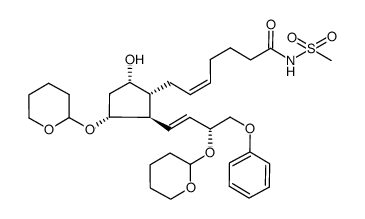 CAS#:64282-81-1
CAS#:64282-81-1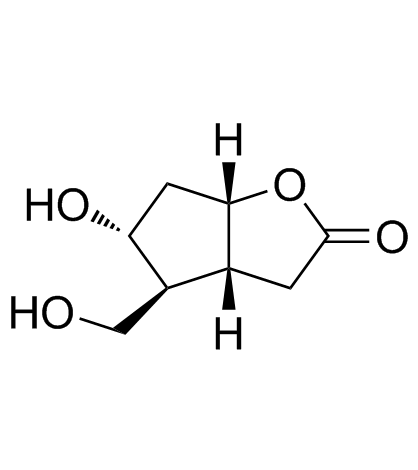 CAS#:32233-40-2
CAS#:32233-40-2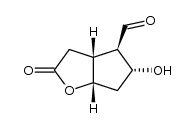 CAS#:62961-72-2
CAS#:62961-72-2![(1S*,5R*,6R*,7R*)-7-trimethylsilyloxy-3-oxo-2-oxabicyclo[3.3.0]octane-6-carbaldehyde Structure](https://www.chemsrc.com/caspic/167/122517-79-7.png) CAS#:122517-79-7
CAS#:122517-79-7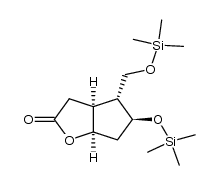 CAS#:118457-23-1
CAS#:118457-23-1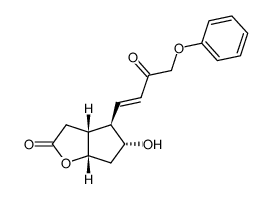 CAS#:871669-32-8
CAS#:871669-32-8