CHEMICAL IDENTIFICATION
-
RTECS NUMBER :
-
OH2915000
-
CHEMICAL NAME :
-
L-Leucine, N-(3-amino-2-hydroxy-1-oxo-4-phenylbutyl)-, (S-(R*,S*))-
-
CAS REGISTRY NUMBER :
-
58970-76-6
-
LAST UPDATED :
-
199612
-
DATA ITEMS CITED :
-
9
-
MOLECULAR FORMULA :
-
C16-H24-N2-O4
-
MOLECULAR WEIGHT :
-
308.42
-
WISWESSER LINE NOTATION :
-
1Y1&1YVQMVYQYZ1R
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
-
TYPE OF TEST :
-
LD - Lethal dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
>2 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968- Volume(issue)/page/year: 36,2971,1983
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
780 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - somnolence (general depressed activity) Skin and Appendages - hair Nutritional and Gross Metabolic - weight loss or decreased weight gain
-
REFERENCE :
-
JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968- Volume(issue)/page/year: 36,2971,1983
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
1900 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - somnolence (general depressed activity) Skin and Appendages - hair Nutritional and Gross Metabolic - weight loss or decreased weight gain
-
REFERENCE :
-
JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968- Volume(issue)/page/year: 36,2971,1983
-
TYPE OF TEST :
-
LD - Lethal dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
>4 gm/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968- Volume(issue)/page/year: 36,2971,1983
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
190 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - somnolence (general depressed activity) Skin and Appendages - hair Nutritional and Gross Metabolic - weight loss or decreased weight gain
-
REFERENCE :
-
JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968- Volume(issue)/page/year: 36,2971,1983
-
TYPE OF TEST :
-
LD50 - Lethal dose, 50 percent kill
-
ROUTE OF EXPOSURE :
-
Subcutaneous
-
SPECIES OBSERVED :
-
Rodent - mouse
-
DOSE/DURATION :
-
1300 mg/kg
-
TOXIC EFFECTS :
-
Behavioral - somnolence (general depressed activity) Skin and Appendages - hair Nutritional and Gross Metabolic - weight loss or decreased weight gain
-
REFERENCE :
-
JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968- Volume(issue)/page/year: 36,2971,1983
-
TYPE OF TEST :
-
LD - Lethal dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Mammal - dog
-
DOSE/DURATION :
-
>1200 mg/kg
-
TOXIC EFFECTS :
-
Details of toxic effects not reported other than lethal dose value
-
REFERENCE :
-
JJANAX Japanese Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo 141, Japan) V.21- 1968- Volume(issue)/page/year: 36,2971,1983 ** OTHER MULTIPLE DOSE TOXICITY DATA **
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Oral
-
SPECIES OBSERVED :
-
Rodent - rat
-
DOSE/DURATION :
-
63700 mg/kg/91D-C
-
TOXIC EFFECTS :
-
Kidney, Ureter, Bladder - proteinuria
-
REFERENCE :
-
OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180, Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 27,401,1984 ** REPRODUCTIVE DATA **
-
TYPE OF TEST :
-
TDLo - Lowest published toxic dose
-
ROUTE OF EXPOSURE :
-
Intraperitoneal
-
DOSE :
-
1250 mg/kg
-
SEX/DURATION :
-
female 16-20 day(s) after conception
-
TOXIC EFFECTS :
-
Reproductive - Effects on Embryo or Fetus - extra-embryonic structures (e.g., placenta, umbilical cord) Reproductive - Effects on Embryo or Fetus - fetotoxicity (except death, e.g., stunted fetus)
-
REFERENCE :
-
HMMRA2 Hormone and Metabolic Research. (Georg Thieme Verlag, Postfach 732, D-7000 Stuttgart 1, Fed. Rep. Ger.) V.1- 1969- Volume(issue)/page/year: 21,366,1989
|
![N-[(2S,3S)-3-azido-2-hydroxy-4-phenylbutanoyl]-L-leucine benzyl ester Structure](https://www.chemsrc.com/caspic/387/121445-53-2.png) CAS#:121445-53-2
CAS#:121445-53-2 CAS#:87304-15-2
CAS#:87304-15-2![benzyl N-[(2S,3R)-3-(N-benzyloxycarbonyl)amino-2-hydroxy-4-phenylbutanoyl]-L-leucinate Structure](https://www.chemsrc.com/caspic/035/60016-66-2.png) CAS#:60016-66-2
CAS#:60016-66-2![methyl N-{(2S,3R)-2-O-benzyl-4-phenyl-3-[9-phenyl-9-fluorenyl-amino]-butanoyl}-L-leucine Structure](https://www.chemsrc.com/caspic/109/577979-03-4.png) CAS#:577979-03-4
CAS#:577979-03-4 CAS#:59969-65-2
CAS#:59969-65-2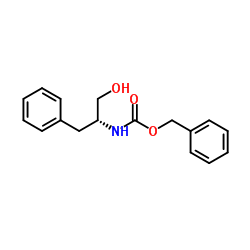 CAS#:58917-85-4
CAS#:58917-85-4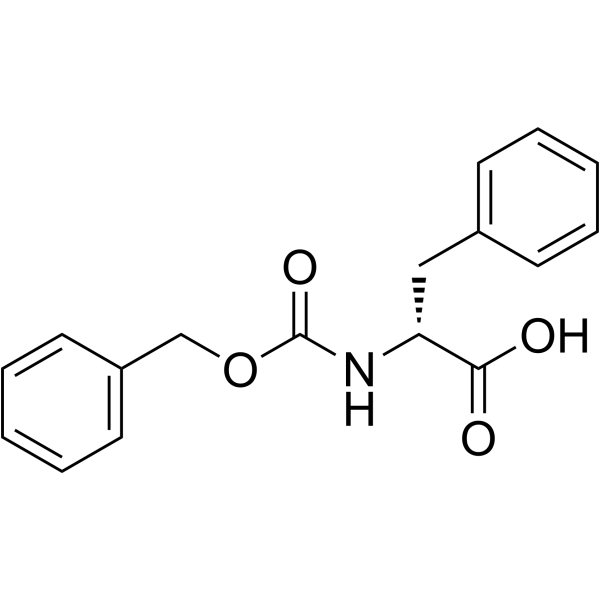 CAS#:2448-45-5
CAS#:2448-45-5![Benzyl [(2R)-1-oxo-3-phenyl-2-propanyl]carbamate Structure](https://www.chemsrc.com/caspic/297/63219-70-5.png) CAS#:63219-70-5
CAS#:63219-70-5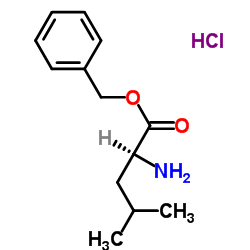 CAS#:2462-35-3
CAS#:2462-35-3