331244-89-4
| Name | (4E)-4-[(2-ethoxyphenyl)hydrazinylidene]-5-methyl-2-(4-phenyl-1,3-thiazol-2-yl)pyrazol-3-one |
|---|---|
| Synonyms |
(4E)-4-[(2-Ethoxyphenyl)hydrazono]-5-methyl-2-(4-phenyl-1,3-thiazol-2-yl)-2,4-dihydro-3H-pyrazol-3-one
S7105,BAX Activator Molecule 7 BAM7 1H-Pyrazole-4,5-dione (3-methyl-1-(4-phenyl-2-thiazolyl) BAX Activator Molecule 7 1H-Pyrazole-4,5-dione, 3-methyl-1-(4-phenyl-2-thiazolyl)-, 4-[2-(2-ethoxyphenyl)hydrazone], (4E)- |
| Description | BAM7 is a direct and selective activator of proapoptotic BAX with an IC50 of 3.3 μM. |
|---|---|
| Related Catalog | |
| Target |
Bax:3.3 μM (IC50) |
| In Vitro | BAM7 is selective for the BH3-binding site on BAX. BAM7 activates BAX and BAX-dependent cell death. Whereas treatment with BAX or BAM7 alone has no effect on the liposomes, the combination of BAM7 and BAX yields dose-responsive liposomal release of entrapped fluorophore. BAM7 dose- and time-responsively impairs the viability of Bak-/- MEFs that exclusively express BAX but has no effect on Bak-/- MEFs that contain BAK but lack BAX. In contrast, standard proapoptotic stimuli such as serum withdrawal, Staurosporine and Etoposide induces an equivalent apoptotic response in Bax-/- and Bak-/- MEFs. As further evidence of BAM7 specificity of action, (i) BAM7 does not affect the viability of Bax-/- Bak-/- MEFs; (ii) ANA-BAM16, which does not bind or activate BAX, has no effect on Bak-/- MEFs; and (iii) BAM7 selectively induces cell death of Bax-/- Bak-/- MEFs reconstituted with wild-type BAX but not BAXK21E , which bears the mutation that abrogates BAM7 binding[1]. |
| Cell Assay | MEF cells are maintained in DMEM high glucose supplemented with 10% (v/v) FBS, 100 U/mL Penicillin, 100 μg/mL Streptomycin, 2 mM L-glutamine, 50 mM HEPES, 0.1 mM MEM nonessential amino acids and 50 μM β-mercaptoethanol. MEFs (2.5×103 cells per well) are seeded in 96-well opaque plates for 18-24 h and then incubated with serial dilutions of BAM7 (3.75, 5, 7.5, 10 and 15 μM), ANA-BAM16 or vehicle (0.15% (v/v) DMSO) in DMEM at 37°C in a final volume of 100 μL. Cell viability is assayed at 24 h by addition of CellTiter-Glo reagent, and luminescence is measured using a SpectraMax M5 microplate reader. Viability assays are performed in at least triplicate, and the data are normalized to vehicle-treated control wells[1]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 567.3±52.0 °C at 760 mmHg |
| Molecular Formula | C21H19N5O2S |
| Molecular Weight | 405.473 |
| Flash Point | 296.9±30.7 °C |
| Exact Mass | 405.125946 |
| PSA | 107.42000 |
| LogP | 3.18 |
| Appearance | light orange to dark orange |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.685 |
| Storage condition | 2-8°C |
| Water Solubility | DMSO: soluble2mg/mL, clear (warmed) |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Hazard Codes | Xn |
| Risk Phrases | 22 |
| RIDADR | NONH for all modes of transport |
|
~% 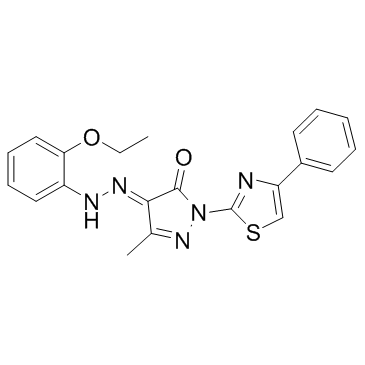
331244-89-4 |
| Literature: DANA FARBER CANCER INSTITUTE, INC.; WALENSKY, Loren D. Patent: WO2013/55949 A2, 2013 ; |
| Precursor 1 | |
|---|---|
| DownStream 0 | |