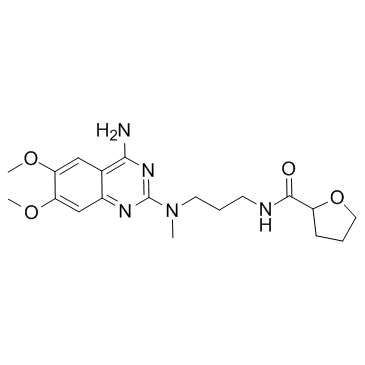
81403-68-1
| Name | alfuzosin hydrochloride |
|---|---|
| Synonyms |
SL 77 499-10
N-{3-[(4-amino-6,7-diméthoxyquinazolin-2-yl)(méthyl)amino]propyl}tétrahydrofurane-2-carboxamide chlorhydrate Uroxatral MFCD00879135 N-{3-[(4-Amino-6,7-dimethoxy-2-quinazolinyl)(methyl)amino]propyl}tetrahydro-2-furancarboxamide hydrochloride (1:1) 2-furancarboxamide, N-[3-[(4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino]propyl]tetrahydro-, monohydrochloride Alfuzosin HCl N-{3-[(4-Amino-6,7-dimethoxychinazolin-2-yl)(methyl)amino]propyl}tetrahydrofuran-2-carboxamidhydrochlorid 2-Furancarboxamide, N-[3-[(4-amino-6,7-dimethoxy-2-quinazolinyl)methylamino]propyl]tetrahydro-, hydrochloride (1:1) SL 77-499-10 Alfuzosin hydrochloride N-{3-[(4-Amino-6,7-dimethoxyquinazolin-2-yl)(methyl)amino]propyl}tetrahydrofuran-2-carboxamide hydrochloride (1:1) Alfuzosin (hydrochloride) |
| Description | Alfuzosin hydrochloride is an α1 adrenergic receptor antagonist used to treat benign prostatic hyperplasia (BPH).Target: α1 adrenergic receptorAlfuzosin, a new quinazoline derivative, acts as a selective and competitive antagonist of alpha 1-adrenoceptor-mediated contraction of prostatic, prostatic capsule, bladder base and proximal urethral smooth muscle, thereby reducing the tone of these structures. Consequently, urethral pressure and resistance, bladder outlet resistance, bladder instability and symptoms associated with benign prostatic hyperplasia are reduced. A limited range of clinical studies have shown oral alfuzosin to be more effective than placebo (in studies of < or = 6 months duration), to have sustained effects on long term administration (< or = 30 months), and to be comparable with the alpha 1-adrenoceptor antagonist prazosin, in the symptomatic treatment of benign prostatic hyperplasia.Oral alfuzosin 7.5 to 10 mg/day in divided doses appears to be a promising first-line agent for symptomatic treatment of noncomplicated mild to moderate benign prostatic hyperplasia in patients with a high dynamic component to their obstruction. In addition, alfuzosin offers an alternative to prostatectomy (the current 'gold standard') in patients who require surgery but are unfit for this treatment, and in patients requiring symptomatic relief while awaiting surgery. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.272 g/cm3 |
|---|---|
| Boiling Point | 687.7ºC at 760 mmHg |
| Melting Point | 225°C |
| Molecular Formula | C19H28ClN5O4 |
| Molecular Weight | 425.910 |
| Exact Mass | 425.182983 |
| PSA | 111.83000 |
| LogP | 3.12480 |
| Storage condition | Store at RT |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Precautionary Statements | P301 + P312 + P330 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn |
| Risk Phrases | R22 |
| RIDADR | NONH for all modes of transport |
|
~89% 
81403-68-1 |
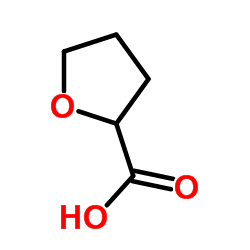
| Literature: WO2008/84493 A2, ; Page/Page column 9-10 ; |
|
~% 
81403-68-1 |
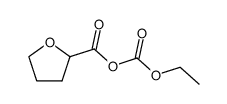
| Literature: WO2009/1369 A1, ; Page/Page column 8 ; |
|
~% 
81403-68-1 |
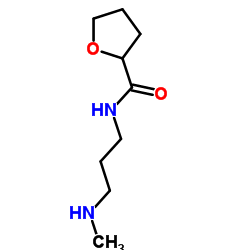
| Literature: US2010/256370 A1, ; Page/Page column 8 ; |
|
~% 
81403-68-1 |
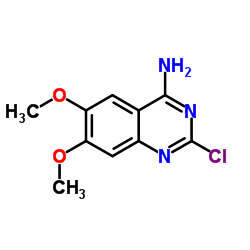
| Literature: Journal of Medicinal Chemistry, , vol. 29, # 1 p. 19 - 25 |
|
~% 
81403-68-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 29, # 1 p. 19 - 25 |
|
~% 
81403-68-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 29, # 1 p. 19 - 25 |
|
~64% 
81403-68-1 |
| Literature: Journal of Medicinal Chemistry, , vol. 29, # 1 p. 19 - 25 |
|
~% 
81403-68-1 |
| Literature: WO2009/7987 A1, ; Page/Page column 10 ; |
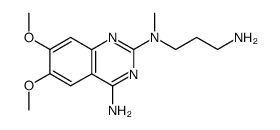
| Precursor 9 | |
|---|---|
| DownStream 1 | |