A-836339
Modify Date: 2024-01-11 00:03:18
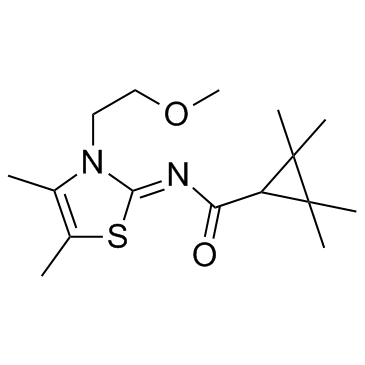
A-836339 structure
|
Common Name | A-836339 | ||
|---|---|---|---|---|
| CAS Number | 959746-77-1 | Molecular Weight | 310.455 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 388.4±44.0 °C at 760 mmHg | |
| Molecular Formula | C16H26N2O2S | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 188.7±28.4 °C | |
Use of A-836339A-836339 is a cannabinoid CB2 receptor-selective agonist; exhibits high potencies at CB(2) and selectivity over CB(1) receptors.IC50 value: 1.6 nM(EC50) [1]Target: CB2 agonistin vitro: In radioligand binding assays, A-836339 displays high affinities at CB(2) receptors and selectivity over CB(1) receptors in both human and rat.In addition A-836339 exhibits a profile devoid of significant affinity at other G-protein-coupled receptors and ion channels [1].in vivo: In the complete Freund's adjuvant model of inflammatory pain, A-836339 exhibits a potent CB(2) receptor-mediated antihyperalgesic effect that is independent of CB(1) or mu-opioid receptors. A-836339 has also demonstrated efficacies in the chronic constrain injury (CCI) model of neuropathic pain, skin incision, and capsaicin-induced secondary mechanical hyperalgesia models [1]. Similar to systemic delivery, intra-spinal injection of A-836339 (0.3 and 1 nmol) also attenuated both von Frey-evoked and spontaneous firing of WDR neurons in neuropathic rats. Intra-spinal injections of A-836339 were ineffective in sham rats [2]. Systemic A-836339 and AM1241 produced dose-dependent efficacy in both inflammatory and neuropathic pain models. Local administration of CB? agonists also produced significant analgesic effects in SNL (intra-DRG and i.t.) and CFA (intra-DRG) pain models [3]. |
| Name | N-[3-(2-methoxyethyl)-4,5-dimethyl-1,3-thiazol-2-ylidene]-2,2,3,3-tetramethylcyclopropane-1-carboxamide |
|---|---|
| Synonym | More Synonyms |
| Description | A-836339 is a cannabinoid CB2 receptor-selective agonist; exhibits high potencies at CB(2) and selectivity over CB(1) receptors.IC50 value: 1.6 nM(EC50) [1]Target: CB2 agonistin vitro: In radioligand binding assays, A-836339 displays high affinities at CB(2) receptors and selectivity over CB(1) receptors in both human and rat.In addition A-836339 exhibits a profile devoid of significant affinity at other G-protein-coupled receptors and ion channels [1].in vivo: In the complete Freund's adjuvant model of inflammatory pain, A-836339 exhibits a potent CB(2) receptor-mediated antihyperalgesic effect that is independent of CB(1) or mu-opioid receptors. A-836339 has also demonstrated efficacies in the chronic constrain injury (CCI) model of neuropathic pain, skin incision, and capsaicin-induced secondary mechanical hyperalgesia models [1]. Similar to systemic delivery, intra-spinal injection of A-836339 (0.3 and 1 nmol) also attenuated both von Frey-evoked and spontaneous firing of WDR neurons in neuropathic rats. Intra-spinal injections of A-836339 were ineffective in sham rats [2]. Systemic A-836339 and AM1241 produced dose-dependent efficacy in both inflammatory and neuropathic pain models. Local administration of CB? agonists also produced significant analgesic effects in SNL (intra-DRG and i.t.) and CFA (intra-DRG) pain models [3]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 388.4±44.0 °C at 760 mmHg |
| Molecular Formula | C16H26N2O2S |
| Molecular Weight | 310.455 |
| Flash Point | 188.7±28.4 °C |
| Exact Mass | 310.171509 |
| PSA | 71.83000 |
| LogP | 3.88 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.561 |
| Storage condition | 2-8℃ |
| Cyclopropanecarboxamide, N-[(2Z)-3-(2-methoxyethyl)-4,5-dimethyl-2(3H)-thiazolylidene]-2,2,3,3-tetramethyl- |
| N-[(2Z)-3-(2-Methoxyethyl)-4,5-dimethyl-1,3-thiazol-2(3H)-ylidene]-2,2,3,3-tetramethylcyclopropanecarboxamide |
| a-836,339 |
| unii-z6y1j258eg |
| A-836339 |