2,5-Thiophenedicarboxaldehyde
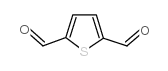
2,5-Thiophenedicarboxaldehyde structure
|
Common Name | 2,5-Thiophenedicarboxaldehyde | ||
|---|---|---|---|---|
| CAS Number | 932-95-6 | Molecular Weight | 140.16000 | |
| Density | 1.37g/cm3 | Boiling Point | 302ºC at 760mmHg | |
| Molecular Formula | C6H4O2S | Melting Point | 114-117ºC | |
| MSDS | Chinese USA | Flash Point | 136.5ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | 2,5-Thiophenedicarboxaldehyde |
|---|---|
| Synonym | More Synonyms |
| Density | 1.37g/cm3 |
|---|---|
| Boiling Point | 302ºC at 760mmHg |
| Melting Point | 114-117ºC |
| Molecular Formula | C6H4O2S |
| Molecular Weight | 140.16000 |
| Flash Point | 136.5ºC |
| Exact Mass | 139.99300 |
| PSA | 62.38000 |
| LogP | 1.37310 |
| Index of Refraction | 1.668 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2934999090 |
|
~41% 
2,5-Thiophenedi... CAS#:932-95-6 |
| Literature: Tetrahedron Letters, , vol. 54, # 22 p. 2795 - 2798 |
|
~0% 
2,5-Thiophenedi... CAS#:932-95-6
Detail
|
| Literature: New Journal of Chemistry, , vol. 26, # 4 p. 373 - 375 |
|
~% 
2,5-Thiophenedi... CAS#:932-95-6 |
| Literature: Tetrahedron Letters, , vol. 35, # 24 p. 4175 - 4176 |
|
~% 
2,5-Thiophenedi... CAS#:932-95-6 |
| Literature: Tetrahedron Letters, , vol. 35, # 24 p. 4175 - 4176 |
|
~% 
2,5-Thiophenedi... CAS#:932-95-6 |
| Literature: Synthesis, , # 4 p. 316 - 318 |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
(Photo)physical Properties of New Molecular Glasses End-Capped with Thiophene Rings Composed of Diimide and Imine Units.
J. Phys. Chem. C Nanomater. Interfaces 118(24) , 13070-13086, (2014) New symmetrical arylene bisimide derivatives formed by using electron-donating-electron-accepting systems were synthesized. They consist of a phthalic diimide or naphthalenediimide core and imine link... |
|
|
Chiral Synthesis via Organoboranes. 46. An Efficient Preparation of Chiral Pyridino- and Thiopheno-18-crown-6 Ligands from Enantiomerically Pure C(2)-Symmetric Pyridine- and Thiophenediols(1).
J. Org. Chem. 64(3) , 721-725, (1999) Asymmetric reduction of 2,6-diacylpyridines with B-chlorodiisopinocampheylborane provides the corresponding C(2)-symmetric diols in very high de and ee. Asymmetric allylboration of 2,6-pyridinedicarbo... |
|
|
Structure-in vitro activity relationships of pentamidine analogues and dication-substituted bis-benzimidazoles as new antifungal agents.
Antimicrob. Agents Chemother. 42(10) , 2495-502, (1998) Twenty analogues of pentamidine, 7 primary metabolites of pentamidine, and 30 dicationic substituted bis-benzimidazoles were screened for their inhibitory and fungicidal activities against Candida alb... |
| thiophene-2,5-dicarbaldehyde |
| MFCD00216592 |