TCMDC-124283
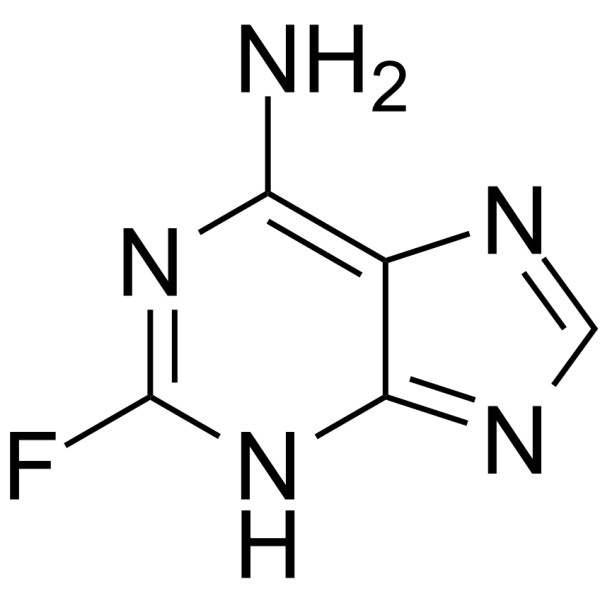
TCMDC-124283 structure
|
Common Name | TCMDC-124283 | ||
|---|---|---|---|---|
| CAS Number | 700-49-2 | Molecular Weight | 153.12 | |
| Density | 1.7±0.1 g/cm3 | Boiling Point | 676.9±58.0 °C at 760 mmHg | |
| Molecular Formula | C5H4FN5 | Melting Point | >350 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 363.2±32.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of TCMDC-1242832-Fluoroadenine is a toxic purine bases. 2-Fluoroadenine has toxicity in nonproliferating and proliferating tumor cells. 2-Fluoroadenine can be used for researching anticancer[1]. |
| Name | 2-fluoroadenine |
|---|---|
| Synonym | More Synonyms |
| Description | 2-Fluoroadenine is a toxic purine bases. 2-Fluoroadenine has toxicity in nonproliferating and proliferating tumor cells. 2-Fluoroadenine can be used for researching anticancer[1]. |
|---|---|
| Related Catalog | |
| Target |
DNA/RNA synthesis[1] |
| In Vitro | 2-Fluoroadenine (2 μM; 4 hours) inhibits CEM cell growth by targeting one or more enzymes involved in either RNA or protein synthesis[1]. 2-Fluoroadenine (0-1000 μM; 96 hours) exhibits cytotoxicity in nonproliferating MRC-5 cells[1]. 2-Fluoroadenine (0.22, 2.2 and 22 μM; 30 hours) inhibits protein, RNA, and DNA syntheses in Balb-3T3 cells incubated in serum-free medium[1]. Cell Proliferation Assay Cell Line: CEM cells[1] Concentration: 2 μM Incubation Time: 4 hours Result: Inhibited CEM cell growth by targeting one or more enzymes involved in either RNA or protein synthesis. Cell Cytotoxicity Assay Cell Line: MRC-5 cells[1] Concentration: 0-1000 μM Incubation Time: 4 hours Result: Exhibited cytotoxicity in nonproliferating MRC-5 cells. |
| References |
| Density | 1.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 676.9±58.0 °C at 760 mmHg |
| Melting Point | >350 °C(lit.) |
| Molecular Formula | C5H4FN5 |
| Molecular Weight | 153.12 |
| Flash Point | 363.2±32.3 °C |
| Exact Mass | 153.045074 |
| PSA | 80.48000 |
| LogP | -0.01 |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C |
| Index of Refraction | 1.783 |
| Storage condition | Refrigerator |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | AU6264700 |
| HS Code | 2933990090 |
|
~% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: WO2006/81665 A1, ; Page/Page column 26-27 ; |
|
~% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: US5180824 A1, ; |
|
~% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: US5180824 A1, ; |
|
~60% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: SAISCHEK, Gerald Patent: US2009/163713 A1, 2009 ; Location in patent: Page/Page column 2 ; |
|
~% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: Nucleosides and Nucleotides, , vol. 18, # 4-5 p. 745 - 757 |
|
~% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: Nucleosides and Nucleotides, , vol. 18, # 4-5 p. 745 - 757 |
|
~% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: Nucleosides and Nucleotides, , vol. 18, # 4-5 p. 745 - 757 |
|
~% 
TCMDC-124283 CAS#:700-49-2 |
| Literature: Nucleosides and Nucleotides, , vol. 18, # 4-5 p. 745 - 757 |
| Precursor 7 | |
|---|---|
| DownStream 3 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Reduced drug incorporation into DNA and antiapoptosis as the crucial mechanisms of resistance in a novel nelarabine-resistant cell line.
BMC Cancer 14 , 547, (2014) Nine-beta-D-arabinofuranosylguanine (ara-G), an active metabolite of nelarabine, enters leukemic cells through human Equilibrative Nucleoside Transporter 1, and is then phosphorylated to an intracellu... |
|
|
Endothelin-1 promotes survival and chemoresistance in chronic lymphocytic leukemia B cells through ETA receptor.
PLoS ONE 9(6) , e98818, (2014) The endothelin axis, comprising endothelins (ET-1, ET-2 and ET-3) and their receptors (ET(A)R and ETBR), has emerged as relevant player in tumor growth and metastasis. Here, we investigated the involv... |
|
|
Preparation of a fludarabine intermediate via selective alkylation of 2-fluoroadenine.
Nucleosides Nucleotides Nucleic Acids 25(7) , 735-45, (2006) The reaction between 2-fluoroadenine (3) and 1,3,5-tri-O-benzyl-1-alpha-D-chloroarabinofuranose (4) with potassium t-amylate was evaluated in various solvents to afford 9-beta-D-(2,3,5-tri-O-benzyl-ar... |
| 2-fluoro-adenin |
| 2-FLUORO-6-AMINOPURINE |
| 7H-Purin-6-amine, 2-fluoro- |
| 2-fluoro-9H-purin-6-amine |
| 2-Fluoroadenine |
| 6-AMINO-2-FLUOROPURINE |
| TCMDC-124283 |
| MFCD01632749 |
| 2-fluoropurine |
| 2-fluoroadenibe |
| 2-Fluoro-1H-purin-6-amine |
| 2-fad |
| 6-amino-2-fluoro-purin |
| BUTTPARK 25 1-01 |
| 2-fluoradenine |
| 2-fluoro-1h-purin-6-amin |
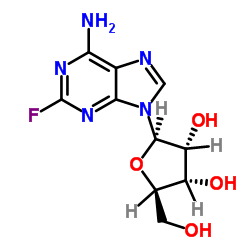
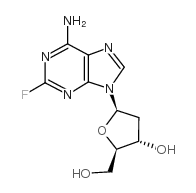
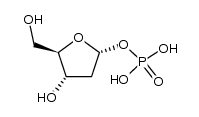
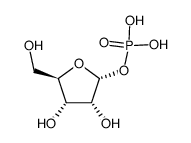
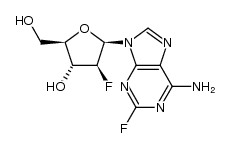
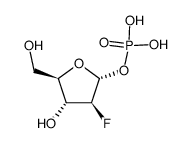
 CAS#:86626-06-4
CAS#:86626-06-4
