Trifluorothymine
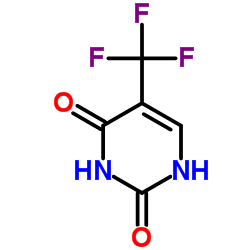
Trifluorothymine structure
|
Common Name | Trifluorothymine | ||
|---|---|---|---|---|
| CAS Number | 54-20-6 | Molecular Weight | 180.085 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 350ºC | |
| Molecular Formula | C5H3F3N2O2 | Melting Point | 245-246 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS06 |
Signal Word | Danger | |
| Name | Trifluorothymine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 350ºC |
| Melting Point | 245-246 °C(lit.) |
| Molecular Formula | C5H3F3N2O2 |
| Molecular Weight | 180.085 |
| Exact Mass | 180.014664 |
| PSA | 65.72000 |
| LogP | 0.04 |
| Index of Refraction | 1.428 |
| Storage condition | −20°C |
| Water Solubility | INSOLUBLE |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301-H315-H319-H335 |
| Precautionary Statements | P261-P301 + P310-P305 + P351 + P338 |
| Hazard Codes | Xi |
| Risk Phrases | R22 |
| Safety Phrases | S24/25 |
| RIDADR | 2811.0 |
| WGK Germany | 3 |
| RTECS | YR1750000 |
| HS Code | 2933599090 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933599090 |
|---|---|
| Summary | 2933599090. other compounds containing a pyrimidine ring (whether or not hydrogenated) or piperazine ring in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Molecular structure and effects of intermolecular hydrogen bonding on the vibrational spectrum of trifluorothymine, an antitumor and antiviral agent.
J. Mol. Model. 18(9) , 4453-64, (2012) In the present work, the experimental and the theoretical vibrational spectra of trifluorothymine were investigated. The FT-IR (400-4000 cm(-1)) and μ-Raman spectra (100-4000 cm(-1)) of trifluorothymi... |
|
|
Quantitative 19F NMR study of trifluorothymidine metabolism in rat brain.
NMR Biomed. 12(6) , 373-80, (1999) Metabolism of trifluorothymidine (TFT) and its transport across the blood-brain barrier (BBB) has been measured quantitatively in rats by fluorine-19 nuclear magnetic resonance spectroscopy ((19)F NMR... |
|
|
Synthesis and in vitro activity of D- and L-enantiomers of 5-(trifluoromethyl)uracil nucleoside derivatives.
Nucleosides Nucleotides Nucleic Acids 20(4-7) , 1123-5, (2001) Recently, beta-L-nucleoside analogues have emerged as a new class of sugar modified nucleosides with potential antiviral and/or antitumoral activity. As a part of our ongoing research on this topic, w... |
| 5-(Trifluoromethyl)-2,4(1H,3H)-pyrimidinedione |
| 5-(trifluoromethyl)-1H-pyrimidine-2,4-dione |
| MFCD00006024 |
| EINECS 200-197-5 |
| 5-(trifluoromethyl)-1,2,3,4-tetrahydropyrimidine-2,4-dione |
| 5-(Trifluoromethyl)uracil |
| 5-(Trifluoromethyl)pyrimidine-2,4(1H,3H)-dione |
| 2,4(1H,3H)-Pyrimidinedione, 5-(trifluoromethyl)- |
 CAS#:2314-97-8
CAS#:2314-97-8 CAS#:66-22-8
CAS#:66-22-8 CAS#:2926-29-6
CAS#:2926-29-6 CAS#:2145-56-4
CAS#:2145-56-4 CAS#:116393-64-7
CAS#:116393-64-7![Bis[trifluoromethyl]mercury Structure](https://image.chemsrc.com/caspic/260/371-76-6.png) CAS#:371-76-6
CAS#:371-76-6![trimethyl-[5-(trifluoromethyl)-2-trimethylsilyloxypyrimidin-4-yl]oxysilane Structure](https://image.chemsrc.com/caspic/180/7057-43-4.png) CAS#:7057-43-4
CAS#:7057-43-4 CAS#:57-13-6
CAS#:57-13-6 CAS#:105480-41-9
CAS#:105480-41-9 CAS#:3932-97-6
CAS#:3932-97-6 CAS#:70-00-8
CAS#:70-00-8 CAS#:65-71-4
CAS#:65-71-4 CAS#:25509-03-9
CAS#:25509-03-9 CAS#:16317-75-2
CAS#:16317-75-2 CAS#:78097-09-3
CAS#:78097-09-3 CAS#:939791-38-5
CAS#:939791-38-5