Cephalexin monohydrate
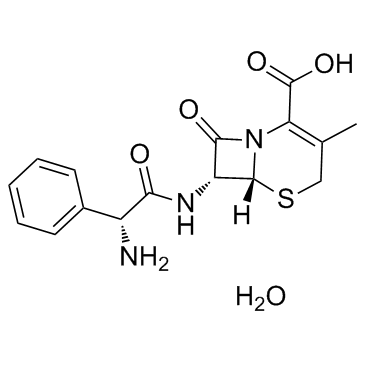
Cephalexin monohydrate structure
|
Common Name | Cephalexin monohydrate | ||
|---|---|---|---|---|
| CAS Number | 23325-78-2 | Molecular Weight | 383.850 | |
| Density | 1.5g/cm3 | Boiling Point | 727.4ºC at 760 mmHg | |
| Molecular Formula | C16H19N3O5S | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 393.7ºC | |
Use of Cephalexin monohydrateCefalexin monohydrate is a cephalosporin antibiotic.Target: AntibacterialCefalexin (INN, BAN) or cephalexin (USAN, AAN) is a first-generation cephalosporin antibiotic introduced in 1967 by Eli Lilly and Company. It is an orally administered agent with a similar antimicrobial spectrum to the intravenous agents cefalotin and cefazolin. It was first marketed as Keflex (Lilly), and is marketed under several other trade names. As of 2008, cefalexin was the most popular cephalosporin antibiotic in the United States, with more than 25 million prescriptions of its generic versions alone, for US$255 million in sales (though less popular than two other antibiotics, amoxicillin and azithromycin, each with 50 million prescriptions per year).Cefalexin is marketed by generic pharmaceutical manufacturers under a wide range of brand names, including: Apo-Cephalex, Biocef, Cefanox, Ceforal, Cephabos, Cephalexin, Cephorum, Ceporex, Cilex, Ialex, Ibilex, Kefexin, Keflet, Keflex, Rekosporin, Keforal, Keftab, Keftal, Lopilexin, Larixin, Novo-Lexin, Ospexin, Tenkorex, Zephalexin, Panixine Disperdose, Rancef, Sialexin, Sporidex and Ulexin. A version of Keflex 750 mg capsules is marketed for twice-daily dosage, to improve compliance. However, it is not a sustained release formulation, and since it is more expensive than the older strengths, some physicians prescribe three 250 mg capsules to be taken twice daily, as a less expensive alternative. |
| Name | cephalexin monohydrate |
|---|---|
| Synonym | More Synonyms |
| Description | Cefalexin monohydrate is a cephalosporin antibiotic.Target: AntibacterialCefalexin (INN, BAN) or cephalexin (USAN, AAN) is a first-generation cephalosporin antibiotic introduced in 1967 by Eli Lilly and Company. It is an orally administered agent with a similar antimicrobial spectrum to the intravenous agents cefalotin and cefazolin. It was first marketed as Keflex (Lilly), and is marketed under several other trade names. As of 2008, cefalexin was the most popular cephalosporin antibiotic in the United States, with more than 25 million prescriptions of its generic versions alone, for US$255 million in sales (though less popular than two other antibiotics, amoxicillin and azithromycin, each with 50 million prescriptions per year).Cefalexin is marketed by generic pharmaceutical manufacturers under a wide range of brand names, including: Apo-Cephalex, Biocef, Cefanox, Ceforal, Cephabos, Cephalexin, Cephorum, Ceporex, Cilex, Ialex, Ibilex, Kefexin, Keflet, Keflex, Rekosporin, Keforal, Keftab, Keftal, Lopilexin, Larixin, Novo-Lexin, Ospexin, Tenkorex, Zephalexin, Panixine Disperdose, Rancef, Sialexin, Sporidex and Ulexin. A version of Keflex 750 mg capsules is marketed for twice-daily dosage, to improve compliance. However, it is not a sustained release formulation, and since it is more expensive than the older strengths, some physicians prescribe three 250 mg capsules to be taken twice daily, as a less expensive alternative. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.5g/cm3 |
|---|---|
| Boiling Point | 727.4ºC at 760 mmHg |
| Molecular Formula | C16H19N3O5S |
| Molecular Weight | 383.850 |
| Flash Point | 393.7ºC |
| Exact Mass | 383.070648 |
| PSA | 147.26000 |
| LogP | 1.40970 |
| Vapour Pressure | 3.27E-22mmHg at 25°C |
| Index of Refraction | 154 ° (C=0.5, H2O) |
| Storage condition | 2-8°C |
| Water Solubility | NH4OH 1 M: 50 mg/mL, clear, yellow |
|
Section 1. Chemical Product and Company Identification Cephalexin, Monohydrate Common Name/ Trade Name Cephalexin, Monohydrate Section 2.Composition and Information on Ingredients Exposure Limits
TWA (mg/m3) STEL (mg/m3) CEIL (mg/m3) % by Weight NameCAS # 1) Cephalexin, Monohydrate23325-78-2100 Toxicological DataNot applicable. on Ingredients Section 3. Hazards Identification Potential Acute HealthSlightly hazardous in case of skin contact (irritant), of eye contact (irritant), of ingestion, of inhalation. Effects CARCINOGENIC EFFECTS: Not available. Potential Chronic Health MUTAGENIC EFFECTS: Not available. Effects TERATOGENIC EFFECTS: Not available. DEVELOPMENTAL TOXICITY: Not available. Repeated or prolonged exposure is not known to aggravate medical condition. Section 4. First Aid Measures Eye ContactCheck for and remove any contact lenses. In case of contact, immediately flush eyes with plenty of water for at least 15 minutes. Cold water may be used. Get medical attention if irritation occurs. Skin ContactWash with soap and water. Cover the irritated skin with an emollient. Get medical attention if irritation develops. Cold water may be used. Serious Skin ContactNot available. InhalationIf inhaled, remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical attention. Serious InhalationNot available. IngestionDo NOT induce vomiting unless directed to do so by medical personnel. Never give anything by mouth to an unconscious person. Loosen tight clothing such as a collar, tie, belt or waistband. Get medical attention if symptoms appear. Serious IngestionNot available. Section 5. Fire and Explosion Data Flammability of the Product May be combustible at high temperature. Auto-Ignition Temperature Not available. Not available. Flash Points Not available. Flammable Limits These products are carbon oxides (CO, CO2), nitrogen oxides (NO, NO2...), sulfur oxides (SO2, SO3...). Products of Combustion Fire Hazards in Presence of Slightly flammable to flammable in presence of heat. Various Substances Risks of explosion of the product in presence of mechanical impact: Not available. Explosion Hazards in Risks of explosion of the product in presence of static discharge: Not available. Presence of Various Substances SMALL FIRE: Use DRY chemical powder. Fire Fighting Media LARGE FIRE: Use water spray, fog or foam. Do not use water jet. and Instructions Special Remarks onMaterial in powder form, capable of creating a dust explosion. Fire Hazards Cephalexin, Monohydrate Special Remarks on Explosion Fine dust dispersed in air in sufficient concentrations, and in the presence of an ignition source is a potential dust explosion hazard. Hazards Section 6. Accidental Release Measures Small SpillUse appropriate tools to put the spilled solid in a convenient waste disposal container. Finish cleaning by spreading water on the contaminated surface and dispose of according to local and regional authority requirements. Large SpillUse a shovel to put the material into a convenient waste disposal container. Finish cleaning by spreading water on the contaminated surface and allow to evacuate through the sanitary system. Section 7. Handling and Storage PrecautionsKeep away from heat. Keep away from sources of ignition. Ground all equipment containing material. Do not ingest. Do not breathe dust. If ingested, seek medical advice immediately and show the container or the label. Keep away from incompatibles such as oxidizing agents. StorageKeep container tightly closed. Keep container in a cool, well-ventilated area. Do not store above 8°C (35.6°F). Refrigerate. Section 8. Exposure Controls/Personal Protection Engineering ControlsUse process enclosures, local exhaust ventilation, or other engineering controls to keep airborne levels below recommended exposure limits. If user operations generate dust, fume or mist, use ventilation to keep exposure to airborne contaminants below the exposure limit. Personal ProtectionSafety glasses. Lab coat. Dust respirator. Be sure to use an approved/certified respirator or equivalent. Gloves. Personal Protection in Case of Splash goggles. Full suit. Dust respirator. Boots. Gloves. A self contained breathing apparatus should be a Large Spillused to avoid inhalation of the product. Suggested protective clothing might not be sufficient; consult a specialist BEFORE handling this product. Exposure LimitsNot available. Section 9. Physical and Chemical Properties Physical state and appearance Solid. (Crystalline powder.)OdorNot available. TasteNot available. 365.41 g/mole Molecular Weight ColorWhite to off-white pH (1% soln/water)Not available. Not available. Boiling Point Melting PointNot available. Not available. Critical Temperature Specific GravityNot available. Vapor PressureNot applicable. Vapor DensityNot available. VolatilityNot available. Not available. Odor Threshold Water/Oil Dist. Coeff.Not available. Not available. Ionicity (in Water) Dispersion PropertiesSee solubility in water. SolubilitySoluble in cold water. Cephalexin, Monohydrate Section 10. Stability and Reactivity Data The product is stable. Stability Instability TemperatureNot available. Excess heat, incompatible materials Conditions of Instability Incompatibility with various Reactive with oxidizing agents. substances Non-corrosive in presence of glass. Corrosivity Special Remarks onNot available. Reactivity Special Remarks onNot available. Corrosivity Will not occur. Polymerization Section 11. Toxicological Information Routes of EntryAbsorbed through skin. Dermal contact. Eye contact. Inhalation. Ingestion. Toxicity to AnimalsAcute oral toxicity (LD50): 1600 mg/kg [Mouse]. Chronic Effects on Humans Not available. Other Toxic Effects onSlightly hazardous in case of skin contact (irritant), of ingestion, of inhalation. Humans Special Remarks onNot available. Toxicity to Animals Special Remarks onMay cause adverse reproductive effects and birth defects (teratogenic). Chronic Effects on Humans Human: passes through the placenta, excreted in maternal milk. Special Remarks on otherAcute Potential Health Effects: Toxic Effects on HumansSkin: May cause skin irritaton. Eyes: May cause respiratory tract irritation. Inhalation: May cause respiratory tract irritation. Ingestion: May cause nausea, vomiting, hypermotility, diarrhea, and decreased appetite. May affect behavior (headache, somnolence, excitement, muscle weakness, ataxia, convulsions), urinary system. May cause tinnitus, diplopia Chronic Potenetial Health Effects: Ingestion: Prologned or repeated ingestion may cause nausea, vomiting,hypermotility, diarrhea. It may affect the urinary system, blood (changes in serum composition, normocytic anemia), adrenal gland, liver. Section 12. Ecological Information Not available. Ecotoxicity Not available. BOD5 and COD Products of BiodegradationPossibly hazardous short term degradation products are not likely. However, long term degradation products may arise. Toxicity of the ProductsThe product itself and its products of degradation are not toxic. of Biodegradation Special Remarks on theNot available. Products of Biodegradation Cephalexin, Monohydrate Section 13. Disposal Considerations Waste DisposalWaste must be disposed of in accordance with federal, state and local environmental control regulations. Section 14. Transport Information DOT ClassificationNot a DOT controlled material (United States). Not applicable. Identification Not applicable. Special Provisions for Transport DOT (Pictograms) Section 15. Other Regulatory Information and Pictograms No products were found. Federal and State Regulations CaliforniaCalifornia prop. 65: This product contains the following ingredients for which the State of California has Proposition 65found to cause cancer which would require a warning under the statute: No products were found. Warnings California prop. 65: This product contains the following ingredients for which the State of California has found to cause birth defects which would require a warning under the statute: No products were found. EINECS: This product is not on the European Inventory of Existing Commercial Chemical Substances. Other Regulations Canada: Not listed on Canadian Domestic Substance List (DSL) or Canadian Non- Domestic Substance List (NDSL). China: Not listed on National Inventory. Japan: Not listed on National Inventory (ENCS). Korea: Not listed on National Inventory (KECI). Philippines: Listed on National Inventory (PICCS). Australia: Listed on AICS. WHMIS (Canada) Not controlled under WHMIS (Canada). Other Classifications This product is not classifiedNot applicable. DSCL (EEC) according to the EU regulations. Health Hazard HMIS (U.S.A.)1 National Fire Protection 1 Flammability 1 Association (U.S.A.) Fire Hazard 1 0 Reactivity Health Reactivity 0 Specific hazard Personal Protection E WHMIS (Canada) (Pictograms) DSCL (Europe) (Pictograms) Cephalexin, Monohydrate TDG (Canada) (Pictograms) ADR (Europe) (Pictograms) Protective Equipment Gloves. Lab coat. Dust respirator. Be sure to use an approved/certified respirator or equivalent. SECTION 16 - ADDITIONAL INFORMATION N/A |
| Hazard Codes | Xn: Harmful; |
|---|---|
| Risk Phrases | R42/43 |
| Safety Phrases | 22-36/37-45 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | XI0350000 |
|
Interaction of Bordetella pertussis filamentous hemagglutinin with human TLR2: identification of the TLR2-binding domain.
APMIS 123(2) , 156-62, (2015) Filamentous hemagglutinin (FHA) is a major adhesion and virulence factor of Bordetella pertussis and also a main component of acellular pertussis vaccines. Interaction of FHA with different receptors ... |
|
|
Disruption of an M. tuberculosis membrane protein causes a magnesium-dependent cell division defect and failure to persist in mice.
PLoS Pathog. 11(2) , e1004645, (2015) The identification of Mycobacterium tuberculosis genes necessary for persistence in vivo provides insight into bacterial biology as well as host defense strategies. We show that disruption of M. tuber... |
|
|
Purification and characterization of the Staphylococcus aureus bacillithiol transferase BstA.
Biochim. Biophys. Acta 1840(9) , 2851-61, (2014) Gram-positive bacteria in the phylum Firmicutes synthesize the low molecular weight thiol bacillithiol rather than glutathione or mycothiol. The bacillithiol transferase YfiT from Bacillus subtilis wa... |
| (6R,7R)-7-{[(2R)-2-Amino-2-phenylacetyl]amino}-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid hydrochloride (1:1) |
| 5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[(2R)-2-amino-2-phenylacetyl]amino]-3-methyl-8-oxo-, (6R,7R)-, hydrochloride (1:1) |
| EINECS 239-773-6 |
| Cephalexin monohydrate |
| MFCD00167148 |
| Cefalexin(compacted) |
| Cefalexin Monohydrate |

