BENZ(C)ACRIDINE
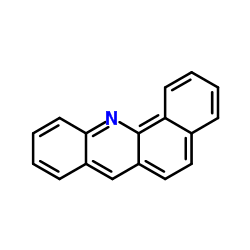
BENZ(C)ACRIDINE structure
|
Common Name | BENZ(C)ACRIDINE | ||
|---|---|---|---|---|
| CAS Number | 225-51-4 | Molecular Weight | 229.276 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 446.2±14.0 °C at 760 mmHg | |
| Molecular Formula | C17H11N | Melting Point | 132°C | |
| MSDS | N/A | Flash Point | 201.4±12.7 °C | |
| Name | benzo[c]acridine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 446.2±14.0 °C at 760 mmHg |
| Melting Point | 132°C |
| Molecular Formula | C17H11N |
| Molecular Weight | 229.276 |
| Flash Point | 201.4±12.7 °C |
| Exact Mass | 229.089142 |
| PSA | 12.89000 |
| LogP | 4.63 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.783 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| RIDADR | UN 2811 |
|---|---|
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2933990090 |
| Precursor 9 | |
|---|---|
| DownStream 1 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Substituted benz[a]acridines and benz[c]acridines as mammalian topoisomerase poisons.
Bioorg. Med. Chem. 8(5) , 1171-82, (2000) Coralyne and several other synthetic benzo[a,g]quinolizium derivatives related to protoberberine alkaloids have exhibited activity as topoisomerase poisons. These compounds are characterized by the pr... |
|
|
Heteroatom effects in chemical carcinogenesis: effects of ring heteroatoms on ease of carbocation formation.
Cancer Biochem. Biophys. 7(1) , 53-60, (1983) The presence of a heteroatom can influence the ease with which a PAH diol-epoxide forms a triol carbocation. The influence of the heteroatom should be greatest when it is located where the PAH undergo... |
|
|
Differentiated genotoxic response of carcinogenic and non-carcinogenic benzacridines and metabolites in rat hepatoma cells.
Carcinogenesis 6(3) , 455-7, (1985) Two closely related hepatoma cell lines were examined for their genotoxic response to benacridines and their metabolites by the appearance of alkaline labile DNA sites: H5, a dedifferentiated line exp... |
| 3,4-Benzacridine |
| Benzo[c]acridine |
| 3,4-Benzoacridine |
| BENZ(C)ACRIDINE |
| 12-Azabenz[a]anthracene |
| Benz[c]acridine |
| 7,8-Benzacridine |
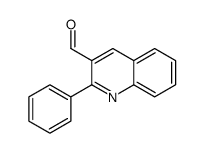 CAS#:227803-21-6
CAS#:227803-21-6![Benzenamine, N-[(2-fluorophenyl)methylene]-2,6-dimethyl- (9CI) Structure](https://image.chemsrc.com/caspic/065/823196-34-5.png) CAS#:823196-34-5
CAS#:823196-34-5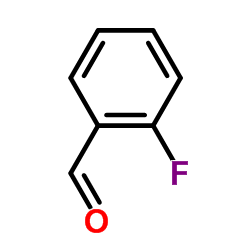 CAS#:446-52-6
CAS#:446-52-6![1,4,7,12-Tetrahydrobenz[c]acridine Structure](https://image.chemsrc.com/caspic/251/77305-61-4.png) CAS#:77305-61-4
CAS#:77305-61-4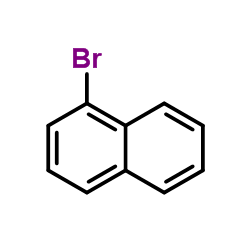 CAS#:90-11-9
CAS#:90-11-9 CAS#:529-23-7
CAS#:529-23-7![1,2,3,4,7,12-Hexahydrobenz[c]acridine Structure](https://image.chemsrc.com/caspic/332/78168-03-3.png) CAS#:78168-03-3
CAS#:78168-03-3 CAS#:563-63-3
CAS#:563-63-3 CAS#:54538-09-9
CAS#:54538-09-9![3,4-dihydrobenzo[c]acridine structure](https://image.chemsrc.com/caspic/088/78167-81-4.png) CAS#:78167-81-4
CAS#:78167-81-4