Docetaxel
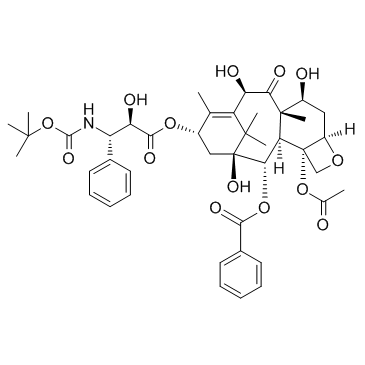
Docetaxel structure
|
Common Name | Docetaxel | ||
|---|---|---|---|---|
| CAS Number | 114977-28-5 | Molecular Weight | 807.879 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 900.5±65.0 °C at 760 mmHg | |
| Molecular Formula | C43H53NO14 | Melting Point | 186-192 °C (dec.) | |
| MSDS | USA | Flash Point | 498.4±34.3 °C | |
Use of DocetaxelDocetaxel is an antineoplastic drug by inhibiting microtubule depolymerization, and attenuating of the effects of bcl-2 and bcl-xL gene expression. |
| Name | docetaxel anhydrous |
|---|---|
| Synonym | More Synonyms |
| Description | Docetaxel is an antineoplastic drug by inhibiting microtubule depolymerization, and attenuating of the effects of bcl-2 and bcl-xL gene expression. |
|---|---|
| Related Catalog | |
| Target |
Microtubule[1] |
| In Vitro | Docetaxel (DOC) and Glufosfamide (GLU) single and combined treatments affect the cells viability in a dose-dependent manner. The IC50 of GLU are 70±4 µM and 86.8±8 µM in PC-3 and LNCaP cells; respectively. While, the IC50 of Docetaxel alone is found to be 3.08±0.4 nM and 1.46±0.2 nM in PC-3 and LNCaP cells; respectively. The co-treatment of GLU with Docetaxel is found to synergize the cytotoxicity and the IC50 values are decreased to be 2.7±0.1 nM and 0.75±0.3 nM in PC-3 and LNCaP cells; respectively[1]. IC50 of NCI-H460 to Docetaxel at 24 h is 116 nM and at 72 h is 30 nM. According to data reported in DTP Data Search, the mean IC50 of NCI-60 cell panel to Docetaxel is 14-34 nM[2]. |
| In Vivo | In female mice, the Docetaxel-induced intestinal apoptosis in the 14-hours after light on (HALO) group is significantly greater than that in the 2-HALO group. Bax expression is significantly elevated by Docetaxel in the 2-HALO group, but not in the 14-HALO group. On the other hand, cleaved Caspase-3 expression is significantly elevated by Docetaxel in the 14-HALO group, but not in the 2-HALO group. The expressions of Wee1 and phosphorylated CKD1 are significantly elevated after dosing of Docetaxel at 14 HALO, but not at 2 HALO. In addition, Docetaxel significantly reduces survivin expression in the 14-HALO group but not in the 2-HALO group. The survivin expression level in the Docetaxel-treated 14-HALO group is significantly smaller than that in the drug-treated 2-HALO group[3]. Piperine (PIP) is administrated via intravenous bolus at 3.5 mg/kg and via oral administration at 35 mg/kg and 3.5 mg/kg, while Docetaxel (DOX) is intravenously administrated at 7 mg/kg to Sprague-Daley rats. The co-administrations of PIP at 35 mg/kg via oral administration and Docetaxel at 7 mg/kg via intravenous bolus administration in Sprague-Dawley rats. The combination use of PIP and Docetaxel results in a synergic increase of both their in vivo exposure[4]. |
| Cell Assay | Single-drug concentration-response curves are assessed. Seeding is done at a density of 2,000 cells/well for PC-3 and LNCaP, in 96-well plates. Cells are treated with each single drug and their combination for 72 h at different drug concentrations. Docetaxel is used at concentrations of 0.1-1,000 nM. GLU is used at concentrations of 0.1-300 µm. Cytotoxicity is assessed at the end of drug exposure using SRB assay. Following 72 h exposure the cells are fixed with 10% trichloroacetic acid (150 µL) for 1 h at 4°C. Then, cells are stained for 10 min at room temperature with 0.4% SRB dissolved in 1% acetic acid. The plates are then air dried for 24 h and the dye is made soluble with 150 µL Tris (10 mM, PH 7.4) for 5 min on a shaker at 1,600 rpm. Absorbance is then measured at 545 nM using microplate reader. Results are expressed as the relative percentage of absorbance compared to control[1]. |
| Animal Admin | Mice[3] Five-week-old male Balb/c mice are used. Docetaxel (0, 10, 20, 30, 40, 60, and 80 mg/kg per week) is given once a week for 3 weeks for mice. Because more than 30 mg/kg per week of Docetaxel causes body weight loss in mice, 20 mg/kg per week of Docetaxel is judged to be the maximum nontoxic dose. Docetaxel (20 mg/kg per week) is given to mice once a week for 3 weeks at one of the following different points (2, 10, 14, or 22 HALO). Seventy-two hours after the final dosing of the agent, the intestinal mucosa of the small intestine (proximal 8 cm) is removed, fixed in 20 N Mildform solution (containing 8% formaldehyde in a buffered solution), and embedded in paraffin blocks, and sections of 5 μm are put on glass slides. Apoptosis is detected using the terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) method, using the Apop Tag Peroxidase In Situ Apoptosis Detection Kit. Rats[4] Male Sprague-Dawley rats with body weight between 230-250 g and age between 6-7 weeks are used. About 25 SD rats are divided into five groups receiving Docetaxel (7 mg/kg, i.v.), PIP (35 mg/kg, p.o.) and their combined administration (DOX+PIP) as well as PIP (3.5 mg/kg, p.o.) and PIP (3.5 mg/kg, i.v.). A day before the drug administrations, the rats are anesthetized with an intramuscular injection of a cocktail containing 60 mg/kg ketamine and 6 mg/kg xylazine (injection volume, 0.2 mL). Right jugular vein is cannulated with a polyethylene tubing (0.5 mm ID, 1 mm) for blood collection. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 900.5±65.0 °C at 760 mmHg |
| Melting Point | 186-192 °C (dec.) |
| Molecular Formula | C43H53NO14 |
| Molecular Weight | 807.879 |
| Flash Point | 498.4±34.3 °C |
| Exact Mass | 807.346619 |
| PSA | 224.45000 |
| LogP | 6.55 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.618 |
| Storage condition | 2~8°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | 26-36/37 |
| RIDADR | 1544 |
| RTECS | DA4172750 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2932999099 |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Androgen receptor (AR) inhibitor ErbB3-binding protein-1 (Ebp1) is not targeted by the newly identified AR controlling signaling axis heat-shock protein HSP27 and microRNA miR-1 in prostate cancer cells.
World J. Urol. 33(3) , 323-7, (2015) Androgen receptor (AR) networks are predominantly involved in prostate cancer (PCa) progression; consequently, factors of AR regulation represent promising targets for PCa therapy. The ErbB3-binding p... |
|
|
Immediate and transient phosphorylation of the heat shock protein 27 initiates chemoresistance in prostate cancer cells.
Oncol. Rep. 32(6) , 2380-6, (2014) Drug resistance minimizes the effects of prostate cancer (PC) chemotherapy with docetaxel and is generally considered to be associated with the expression of heat shock protein (HSP) 27 including vari... |
|
|
Inhibition of PI3Kβ signaling with AZD8186 inhibits growth of PTEN-deficient breast and prostate tumors alone and in combination with docetaxel.
Mol. Cancer Ther. 14(1) , 48-58, (2015) Loss of PTEN protein results in upregulation of the PI3K/AKT pathway, which appears dependent on the PI3Kβ isoform. Inhibitors of PI3Kβ have potential to reduce growth of tumors in which loss of PTEN ... |
| (5β,7β,10β,13α)-4-Acetoxy-13-({(2R,3S)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl}oxy)-1,7,10-trihydroxy-9-oxo-5,20-epoxytax-11-en-2-yl benzoate |
| Benzenepropanoic acid, β-[[(1,1-dimethylethoxy)carbonyl]amino]-α-hydroxy-, (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6,11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester, (αR,βS)- |
| Benzenepropanoic acid, β-[[(1,1-dimethylethoxy)carbonyl]amino]-α-hydroxy-, (2aR,4S,4aS,6R,9S,11S,12S,12aR,12bS)-12b-(acetyloxy)-12-(benzoyloxy)-2a,3,4,4a,5,6,9,10,11,12,12a,12b-dodecahydro-4,6, 11-trihydroxy-4a,8,13,13-tetramethyl-5-oxo-7,11-methano-1H-cyclodeca[3,4]benz[1,2-b]oxet-9-yl ester, (αR,βS)- |
| D-2-amino-5-phosphonovaleric acid |
| APV |
| AP-5 |
| DL-APV |
| (2α,5β,7β,10β,13α)-4-Acetoxy-13-({(2R,3S)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl}oxy)-1,7,10-trihydroxy-9-oxo-5,20-epoxytax-11-en-2-yl benzoate |
| MFCD00800737 |
| DOCETAXEL TRIHYDRATE |
| Docetaxol |
| 5-PHOSPHONO-DL-NORVALINE |
| Taxotere |
| (2α,5β,7β,10β,13α)-4-Acetoxy-1,7,10-trihydroxy-13-{[(2R,3S)-2-hydroxy-3-({[(2-methyl-2-propanyl)oxy]carbonyl}amino)-3-phenylpropanoyl]oxy}-9-oxo-5,20-epoxytax-11-en-2-yl benzoate |
| DL-2-amino-5-phosphonovaleric acid |
| DOCETAXEL(TAXOTERE) |
| N-Debenzoyl-N-tert-butoxycarbonyl-10-deacetyltaxol |
| Docetaxel |
| N-DEBENZOYL-N-TERT-BUTOXYCARBONYL-10-DEACETYL |
| DOCETAXEL(TAXOTERE) UK 427857 |
| N-debenzoyl-N-Boc-10-deacetyl taxol |
| TAXOTERE, DOCETAXEL |
| (2α,5β,7β,10β,13α)-4-(acetyloxy)-13-({(2R,3S)-3-[(tert-butoxycarbonyl)amino]-2-hydroxy-3-phenylpropanoyl}oxy)-1,7,10-trihydroxy-9-oxo-5,20-epoxytax-11-en-2-yl benzoate |
| 2-amino-5-phosphopentanoicacid |
| N-debenzoyl-N-tert-butoxycarbonyl-10-deacetyl taxol |
| 2-amino-5-phosphonovaleric acid |
| acide amino-6 carboxy-6 hexylphosphonique |
| 2-amino-5-phosphovaleric acid |

