37517-30-9
| Name | acebutolol |
|---|---|
| Synonyms |
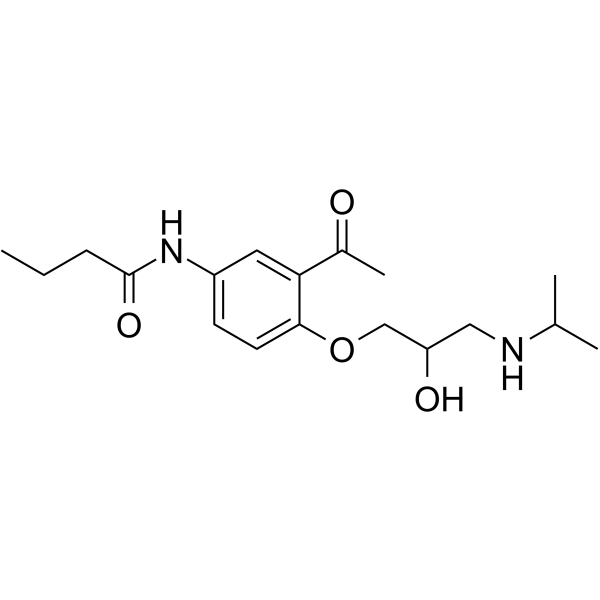
3'-Acetyl-4'-[2-hydroxy-3-(isopropylamino)propoxy]butyranilide
アセブトロール EINECS 253-539-0 N-{3-acetyl-4-[2-hydroxy-3-(propan-2-ylamino)propoxy]phenyl}butanamide N-{3-Acetyl-4-[2-hydroxy-3-(isopropylamino)propoxy]phenyl}butanamide acebutolol N-(3-Acetyl-4-(2-hydroxy-3-(isopropylamino)propoxy)phenyl)butyramide IL 17803A MFCD00599435 Acecor |
| Description | Acebutolol is an orally active β1 adrenergic receptor (β1AR) antagonist. Acebutolol is used for hypertension, angina pectoris and cardiac arrhythmias research[1][2][3]. |
|---|---|
| Related Catalog | |
| Target |
β1 adrenoceptor |
| In Vivo | Acebutolol is a beta blocker for the treatment of hypertension and arrhythmias. Acebutolol following single intravenous administration (10 mg/kg) to rat results in the plasma clearance of 61.9 mL/min/kg, the volume of distribution of 9.6 L/kg, and an elimination half-life of 1.8 hours. Acebutolol following single intravenous administration (50 mg/kg) to rat results in the plasma clearance of 46.5 mL/min/kg, the volume of distribution of 9.5 L/kg, and an elimination half-life of 2.3 hours[1]. Acebutolol (30 mg/kg) decreases cardiac output by 65% and 31% after 1 min and 10 min measurements, respectively, in Sprague-Dawley rats. Acebutolol (30 mg/kg) significantly reduces regional blood flow (RBF) in most organs either after 1 min or 10 min measurements when compare with the baseline values in Sprague-Dawley rats[3]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 564.1±50.0 °C at 760 mmHg |
| Melting Point | 119-123ºC |
| Molecular Formula | C18H28N2O4 |
| Molecular Weight | 336.426 |
| Flash Point | 295.0±30.1 °C |
| Exact Mass | 336.204895 |
| PSA | 87.66000 |
| LogP | 1.95 |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.543 |
| Water Solubility | 259 mg/L |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| HS Code | 2924299090 |
|---|
| HS Code | 2924299090 |
|---|---|
| Summary | 2924299090. other cyclic amides (including cyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |