944808-88-2
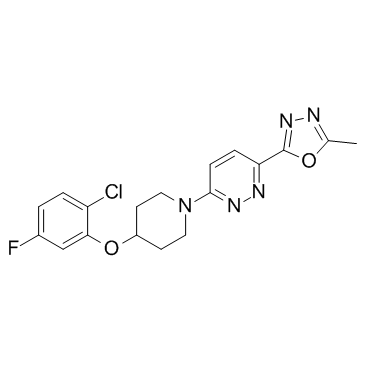
| Name | 2-[6-[4-(2-chloro-5-fluorophenoxy)piperidin-1-yl]pyridazin-3-yl]-5-methyl-1,3,4-oxadiazole |
|---|---|
| Synonyms |
3-[4-(2-CHLORO-5-FLUOROPHENOXY)-PIPERIDIN-1-YL]-6-(5-METHYL-1,3,4-OXADIAZOL-2-YL)-PYRIDAZINE
(3-[4-(2-chloro-5-fluorophenoxy)-1-piperidinyl]-6-(5-methyl-1,3,4-oxadiazol-2-yl)-pyridazine) 3-[4-(2-chloro-5-fluorophenoxy)piperidin-1-yl]-6-(5-methyl-1,3,4-oxadiazol-2-yl)pyridazine 3-[4-(2-Chloro-5-fluorophenoxy)-1-piperidinyl]-6-(5-methyl-1,3,4-oxadiazol-2-yl)pyridazine CAY10566 |
| Description | CAY10566 is a stearoyl-CoA desaturase (SCD) inhibitor. |
|---|---|
| Related Catalog | |
| Target |
SCD[1] |
| In Vitro | Treatment with specific SCD inhibitor CAY10566 dose-dependently reduces SCD activity in MOVAS-1 cells resulting in a significant increase in endoplasmic reticulum (ER) stearate levels. SCD inhibition by CAY10566 treatment induces mineralization and osteoblastic differentiation of MOVAS-1 cells. Along with induction of vascular calcification, CAY10566 dose-dependently induces total ATF4, p-ATF4, and p-eIF2α protein expression. ATF4 mRNA, CHOP protein, CHOP mRNA, and sXBP-1 mRNA levels are also highly and dose-dependently induced by CAY10566 treatment. Phosphorylated PKR-like endoplasmic reticulum kinase (PERK) levels are increased by 15.7-fold at 2 h of CAY10566 treatment, whereas p-eIF2α levels are transiently increased by 1.91-fold at 6 h of CAY10566 treatment. The expressions of ATF4, CHOP, and sXBP mRNA are induced up to 96 h of 300 nM CAY10566 treatment[1]. |
| In Vivo | After establishment of palpable tumors, the mice are treated with vehicle or SCD1 inhibitor (2.5 mg/kg CAY10566 orally twice daily). The effect of SCD1 inhibition on the Akt-driven tumors is greater than on the Ras-driven tumors, with the mean tumor volume at day 13 or 14 post therapy, relative to untreated tumors, 0.5±0.04 and 0.67±0.05 respectively (P=0.01 for Ras-Akt comparison, by two-tailed t test)[2]. |
| Cell Assay | MOVAS-1 cells treated with CAY10566 are incubated with 200 μM stearate-BSA complex containing 1 μCi 14C-stearate. Total lipids are saponified with 3 M sodium hydroxide/ethanol. The saponified fatty acids are separated by 10% silver nitrate-coated thin-layer chromatography. The ratio of the cpm in the band corresponding to oleic acid to the cpm in the band corresponding to stearate is used to calculate stearoyl-CoA desaturase (SCD) activity as previously described[1]. |
| Animal Admin | To generate allografts, 1×107 immortalized baby mouse kidney (iBMK) cells are implanted in Matrigel into nu/nu athymic female mice. After establishment of palpable tumors, mice are randomized to receive 2.5 mg/kg CAY10566 orally twice daily in 0.5% methylcellulose or vehicle control. Xenograft tumors are measured biweekly and tumor volume calculated as volume=(length×width2×π)/6[2]. |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 600.2±65.0 °C at 760 mmHg |
| Molecular Formula | C18H17ClFN5O2 |
| Molecular Weight | 389.811 |
| Flash Point | 316.8±34.3 °C |
| Exact Mass | 389.105469 |
| PSA | 77.17000 |
| LogP | 1.88 |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.592 |
| Storage condition | 2-8℃ |
| Precursor 2 | |
|---|---|
| DownStream 0 | |