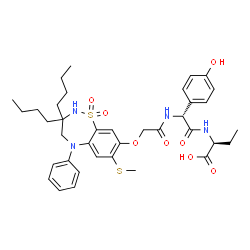
501692-44-0
| Name | odevixibat |
|---|---|
| Synonyms |
odevixibat
(2S)-2-{[(2R)-2-[({[3,3-Dibutyl-7-(methylsulfanyl)-1,1-dioxido-5-phenyl-2,3,4,5-tetrahydro-1,2,5-benzothiadiazepin-8-yl]oxy}acetyl)amino]-2-(4-hydroxyphenyl)acetyl]amino}butanoic acid |
| Description | A4250 (A-4250, Odevixibat) is a small molecule ileal bile acid transporter (IBAT) inhibitor for the treatment of cholestatic liver diseases including progressive familial intrahepatic cholestasis and NASH. Other Indication Phase 3 Clinical |
|---|---|
| Related Catalog | |
| Target |
IC50: IBAT[1] |
| In Vivo | Odevixibat (A4250)(0.01% (w/w) in chow diet; 4 weeks) improves sclerosing cholangitis and significantly reduces serum alanine aminotransferase, alkaline phosphatase and BAs levels, hepatic expression of pro-inflammatory and pro-fibrogenic genes and bile duct proliferation in Mdr2-/- mice[1]. In addition, Odevixibat (A4250) significantly reduces bile flow and biliary BA output, which correlates with reduced bsep transcription, while Ntcp and Cyp7a1 are induced[1]. Animal Model: Eight week old Mdr2-/- (Abcb4-/-) mice (model of cholestatic liver injury and sclerosing cholangitis)[1] Dosage: 0.01% (w/w) in chow diet Administration: 4 weeks Result: Decreased cholestatic liver and bile duct injury in mice model. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Molecular Formula | C37H48N4O8S2 |
| Molecular Weight | 740.929 |
| Exact Mass | 740.291382 |
| LogP | 7.03 |
| Index of Refraction | 1.643 |