TGR5
Modify Date: 2025-08-27 16:00:32

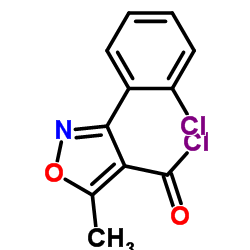
TGR5 structure
|
Common Name | TGR5 | ||
|---|---|---|---|---|
| CAS Number | 1197300-24-5 | Molecular Weight | 361.222 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 541.8±50.0 °C at 760 mmHg | |
| Molecular Formula | C18H14Cl2N2O2 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 281.5±30.1 °C | |
Use of TGR5TGR5 Receptor Agonist, a potent TGR5(GPCR19) agonist, showed improved potency in the U2-OS cell assay (pEC50 = 6.8) and in melanophore cells (pEC50 = 7.5).IC50 value: 6.8 (pEC50, U2-OS cell assay); 7.5(pEC50, melanophore cell) [1]Target: TGR5TGR5 Receptor Agonist was profiled against more than 100 in-house and external 7TM, ion channel, enzyme, transporter, and nuclear hormone receptor selectivity assays, including FXR, another bile acid receptor, and showed significant response only in secretion of the pro-inflammatory cytokine TNFalpha (pIC50 = 6.8) in human primary monocytes following stimulation with LPS (lipopolysaccharide). In addition, TGR5 Receptor Agonist has good physicochemical properties and no measurable activity against three of the common cytochrome P450 (CYP450) isoforms (1A2, 2C9, and 2D6) or hERG dofetilide binding (pIC50 <4.3). In rat pharmacokinetic (PK) studies, however, TGR5 Receptor Agonist showed high in vivo clearance (Cl = 85 mL/min/kg) and intrinsic clearance (Clint = 48 mL/min/g) which provided a reasonable explanation for the observed poor exposure. Because the TGR5 receptor is expressed in the GI tract at levels that increase corresponding with L-cell population density, we believe that agonists such as 6 and 7 possessing poor systemic exposure are good tool compounds for directly targeting the TGR5 receptor in the GI tract via local administration (vide infra) rather than systemic exposure. Our hypothesis was that for this receptor, systemic exposure was not necessary to achieve the desired effect of stimulating GLP-1 secretion in vivo [1]. |
| Name | 3-(2-Chlorophenyl)-N-(4-chlorophenyl)-N,5-dimethyl-1,2-oxazole-4- carboxamide |
|---|---|
| Synonym | More Synonyms |
| Description | TGR5 Receptor Agonist, a potent TGR5(GPCR19) agonist, showed improved potency in the U2-OS cell assay (pEC50 = 6.8) and in melanophore cells (pEC50 = 7.5).IC50 value: 6.8 (pEC50, U2-OS cell assay); 7.5(pEC50, melanophore cell) [1]Target: TGR5TGR5 Receptor Agonist was profiled against more than 100 in-house and external 7TM, ion channel, enzyme, transporter, and nuclear hormone receptor selectivity assays, including FXR, another bile acid receptor, and showed significant response only in secretion of the pro-inflammatory cytokine TNFalpha (pIC50 = 6.8) in human primary monocytes following stimulation with LPS (lipopolysaccharide). In addition, TGR5 Receptor Agonist has good physicochemical properties and no measurable activity against three of the common cytochrome P450 (CYP450) isoforms (1A2, 2C9, and 2D6) or hERG dofetilide binding (pIC50 <4.3). In rat pharmacokinetic (PK) studies, however, TGR5 Receptor Agonist showed high in vivo clearance (Cl = 85 mL/min/kg) and intrinsic clearance (Clint = 48 mL/min/g) which provided a reasonable explanation for the observed poor exposure. Because the TGR5 receptor is expressed in the GI tract at levels that increase corresponding with L-cell population density, we believe that agonists such as 6 and 7 possessing poor systemic exposure are good tool compounds for directly targeting the TGR5 receptor in the GI tract via local administration (vide infra) rather than systemic exposure. Our hypothesis was that for this receptor, systemic exposure was not necessary to achieve the desired effect of stimulating GLP-1 secretion in vivo [1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 541.8±50.0 °C at 760 mmHg |
| Molecular Formula | C18H14Cl2N2O2 |
| Molecular Weight | 361.222 |
| Flash Point | 281.5±30.1 °C |
| Exact Mass | 360.043243 |
| PSA | 46.34000 |
| LogP | 3.30 |
| Appearance of Characters | white solid |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.627 |
| InChIKey | IGRCWJPBLWGNPX-UHFFFAOYSA-N |
| SMILES | Cc1onc(-c2ccccc2Cl)c1C(=O)N(C)c1ccc(Cl)cc1 |
| Storage condition | -20℃ |
| Precursor 2 | |
|---|---|
| DownStream 0 | |
Total 83, Current Page 1 of 9
1
2
3
4
5
| Ether,heptafluoropropyl vinyl |
| TGR5 Receptor Agonist |
| 3-(2-Chlorophenyl)-N-(4-chlorophenyl)-N,5-dimethyl-1,2-oxazole-4-carboxamide |
| heptafluoropropyl vinyl ether |
| Perfluoro propyl vinyl ether |
| CCDC |
| TGR5 |