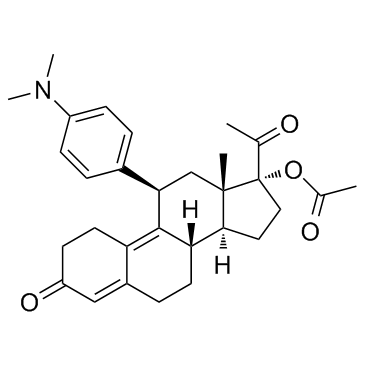
Ulipristal acetate
Published date: 2022-08-10 10:48:52
| Name | Ulipristal acetate | CAS# | 126784-99-4 | |
|---|---|---|---|---|
| Price | $10000.0/1kg | Purity | 99.5% | |
| Stocking Period |
Inquiry | Stock | In Stock | |
| Product Webpage: | http://www.yueweichem.com/products_detail.php?cid=7&id=372 | |||
| Detail
Product Information
Product name: 17α-Acetoxy-11β-[4-(N,N-dimethylamino)phenyl]-19-norpregna-4,9-diene-3,20-dione CAS number: 126784-99-4 Molecular Formula C30H37NO4 Molecular Weight 475.62 Appearance:White or light yellow crystalline powder |
Supplier/Manufacture
Jinan Yuewei Chemical Co.,ltd
Chemsrc Level: UnverifiedTel: 86-13583181986
Address: 9th Floor North,Office Building of Jinan New Material Trade Center
Area: China
Contact: meng
Contact Phone #: 86-13583181986
Email: meng@yueweichemical.com
Website: http://www.yueweichem.com
Major Market: Europe,Korea,Brazil
Annual Trade Volume:
Foreign Trade % of Sales:
Major partners:
Jinan Yuewei Chemical Co.,Ltd is located in Jinan New Material Industrial Park,which is a provincial development zone approved by Government. We engage in researching, producing and selling Active Pharmaceutical Ingredients including antineoplastic, anticoagulant, antiallergic(ophthalmological), advanced intermediates and fine chemicals (eg.Fluoride Series,food additives).
Our APIs are equipped with GMP certificate;fine chemicals are produced in factories certificated with ISO9001,both of which insure the traceability,standardization and high quality.
Our customers include worldwide leading pharmaceutical and chemical companies, trustworthy distributors,universities, research institutes; we consider them as partners; we keep providing qualified products, competitive prices and excellent service. In order to meet various requirements, both bulk quantity and small packing level like gram, kilogram are available.
With more than 10 years experience, we have been a reliable, experienced and creative enterprise .Our products regularly export to Europe,Brazil,Pakistan,India and other areas. The quality and services are well received by customers.
Our company's principle is:"Honesty, Efficiency, Mutual benefit". We sincerely hope to cooperate with customers all over the world.
Our APIs are equipped with GMP certificate;fine chemicals are produced in factories certificated with ISO9001,both of which insure the traceability,standardization and high quality.
Our customers include worldwide leading pharmaceutical and chemical companies, trustworthy distributors,universities, research institutes; we consider them as partners; we keep providing qualified products, competitive prices and excellent service. In order to meet various requirements, both bulk quantity and small packing level like gram, kilogram are available.
With more than 10 years experience, we have been a reliable, experienced and creative enterprise .Our products regularly export to Europe,Brazil,Pakistan,India and other areas. The quality and services are well received by customers.
Our company's principle is:"Honesty, Efficiency, Mutual benefit". We sincerely hope to cooperate with customers all over the world.
Top Suppliers:
- Shanghai Nianxing Industrial Co., Ltd
- China
- Product Name: Ulipristal acetate
- Price: Check
- Purity: 98.0%
- Stocking Period: 10 Day
- Contact: Yang
- Email: sales@echemcloud.com
- Dayang Chem (Hangzhou) Co., Ltd.
- China
- Product Name: Ulipristal-acetate
- Price: $Inquiry/100g $Inquiry/1kg $Inquiry/100kg $Inquiry/1000kg
- Purity: 98.0%
- Stocking Period: Inquiry
- Contact: Ms Wang
- Email: enquiry@dycnchem.com
- Shanghai Amole Biotechnology Co., Ltd.
- China
- Product Name: Ulipristal acetate
- Price: ¥Inquiry/100mg ¥Inquiry/10mg ¥Inquiry/1g ¥Inquiry/250mg
- Purity: 98.0%
- Stocking Period: Inquiry
- Contact: Xiyao Wang
- Email: 3623107365@qq.com
- Shanghai Jizhi Biochemical Technology Co., Ltd
- China
- Product Name: Ulipristal acetate
- Price: ¥1038.0/250mg
- Purity: 98.0%
- Stocking Period: 2 Day
- Contact: Liu jia


- Email: 2130147988@qq.com
- Henan Tianfu Chemical Co., Ltd.
- China
- Product Name: Ulipristal Acetate
- Price: $Inquiry/1kg $Inquiry/25kg $Inquiry/1000kg $Inquiry/10ton
- Purity: 99.0%
- Stocking Period: Inquiry
- Contact: Anson
- Email: info@tianfuchem.com
Get all suppliers by the below link:
Price: $66/10mM*1mLinDMSO
Price from the other suppliers:
| 2024-07-15 |
Ulipristal acetate
Price: Inquiry |
| 2021-07-26 |
Ulipristal-acetate
Price: $Inquiry |
| 2019-03-05 |
Ulipristal acetate
Price: ¥1038.0 |
| 2025-09-17 |
Ulipristal acetate
Price: ¥Inquiry |
| 2024-01-09 |
Ulipristal Acetate
Price: $Inquiry |
| 2017-06-12 |
Ulipristal acetate
Price: ¥Inquiry |
| 2018-03-26 |
Ulipristal acetate
Price: ¥Inquiry |
| 2022-03-22 |
Ulipristal Acetate
Price: $Inquiry |
| 2024-04-24 |
Ulipristal acetate
Price: ¥Inquiry |
Get all price from the following link:
Related product price:
- 3-(2,4-Dimethylphenyl)azetidine price
- N-(2-Methoxy-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin-3-yl)-4-(trifluoromethyl)benzenesulfonamide price
- Methyl[2-(2-methylphenyl)propan-2-yl]amine price
- 5-Oxazolamine, 2-(2-thienyl) price
- 2-(3,4-Dimethoxyphenyl)cyclopropanecarbonitrile price
- 4-(1-Isocyanatocyclopropyl)-1-methoxy-2-methylbenzene price
- 2-(1-Isocyanatocyclopropyl)-1-methoxy-4-methylbenzene price
- N-(3,5-Dimethylphenyl)succinimide price
- 2-(5-Bromo-1,3,4-oxadiazol-2-yl)ethan-1-ol price
- 5-(2-phenoxyethyl)-1H-pyrazol-3-amine price
Ulipristal acetate price is $10000.0/1kg. Find complete details about Ulipristal acetate product information includes price/quote, purity, stocking period etc. from Jinan Yuewei Chemical Co.,ltd>>Mobile(AMP)
version: Ulipristal acetate Price from
Supplier Brand Jinan Yuewei Chemical Co.,ltd