| Structure | Name/CAS No. | Articles |
|---|---|---|
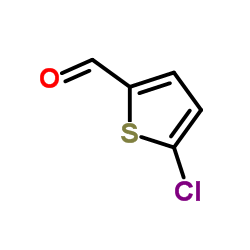 |
5-Chloro-2-thiophenecarbaldehyde
CAS:7283-96-7 |
M da Consolação, F Linardi, M M de Oliveira, M R Sampaio
Index: J. Med. Chem. 18(11) , 1159-61, (1975)
Full Text: HTML
Lapachol [2-hydroxy-3-(3-methyl-2-butenyl)-1,4-naphthoquinone] and its analogs [2-(3,7-dimethyl-2,6-octadienyl)-3-hydroxy-1,4-naphthoquinone and 2-(3,3-dibromo-2-propenyl)-3-hydroxy-1,4-naphthoquinone] have been described, among almost a hundred synthesized analogs, as active against rat tumor Walker 256 carcinosarcoma. The acetylglucosylation of lapachol results in a compound which extends lapachol activity becoming effective against mouse lymphocytic leukemia P-388. When mice inoculated with 10(6) leukemic cells were treated with the drug during 9 days, their life span increased 80% over the control animals. Identification spectral data (uv, ir, 1H NMR, and MS) of the compound obtained by synthesis are given.
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
 |
5-Chloro-2-thiophenecarbaldehyde
CAS:7283-96-7 |
C5H3ClOS |
|
Synthesis and antiinflammatory activity of some 2-heteroaryl...
1975-11-01 [J. Med. Chem. 18(11) , 1158-9, (1975)] |
|
N, N'-Bis [(E)-(5-chloro-2-thienyl) methyliden...
[Acta Crystallogr. Sect. E Struct. Rep. Online 66(11) , o2883, (2010)] |
|
(E)-4-[(5-Chloro-2-thienyl) methyleneamino]-1, 5-dime...
[Acta Crystallogr. Sect. E Struct. Rep. Online 63(2) , o912-o913, (2007)] |
|
Synthesis of 6-(Thienylmethylene)penam Sulfones and their ß-...
[Yakhak Hoechi 51(6) , (2007)] |
Home | MSDS/SDS Database Search | Journals | Product Classification | Biologically Active Compounds | Selling Leads | About Us | Disclaimer
Copyright © 2026 ChemSrc All Rights Reserved