Action of brassinosteroids on the epithelial cell line from Chironomus tentans.
K D Spindler, M Spindler-Barth, A Turberg, G Adam
Index: Z. Naturforsch., C, J. Biosci. 47(3-4) , 280-4, (1992)
Full Text: HTML
Abstract
The two brassinosteroids, 22 S,23 S-homobrassinolide and 22 S,23 S-homocastasterone are weak competitors of the binding of [3H]ponasterone A to the intracellular ecdysteroid receptor from the epithelial cell line from Chironomus tentans. The relative affinities to the ecdysteroid receptor are 0.001 for both brassinosteroids as compared to 20-OH-ecdysone and 0.1 in comparison to ecdysone. Both substances exert morphological effects similar to those observed with 20-OH-ecdysone. Like moulting hormones both brassinosteroids inhibit chitin synthesis. However, these effects were observed only at rather high concentrations (10(-5) to 10(-4) M) which were cytotoxic for 22 S,23 S-homobrassinolide.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
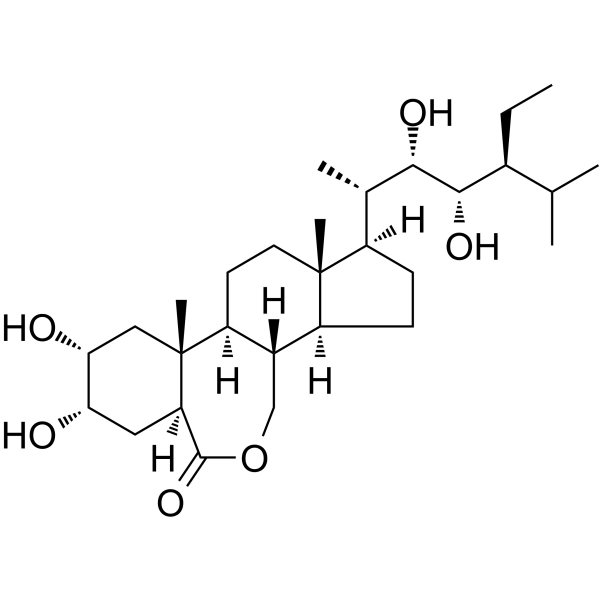 |
Isohomobrassinolide
CAS:80483-89-2 |
C29H50O6 |
|
Akt-dependent anabolic activity of natural and synthetic bra...
2011-06-23 [J. Med. Chem. 54 , 4057-4066, (2011)] |
|
Brassinosteroids protect Lycopersicon esculentum from cadmiu...
2010-03-01 [Protoplasma 239(1-4) , 3-14, (2010)] |
|
Synthesis and bioactivity of natural and C-3 fluorinated bio...
2001-11-01 [Phytochemistry 58(6) , 973-80, (2001)] |
|
Synthesis and biological activity of furostanic analogues of...
2007-12-01 [Steroids 72(14) , 955-9, (2007)] |
|
Studies on steroidal plant-growth regulators (XVII)--a new s...
1991-06-01 [Sci. China,. Ser. B, Chem. Life Sci. Earth Sci. 34(6) , 706-11, (1991)] |