Synthesis of (+/-)- and (-)-vibralactone and vibralactone C.
Quan Zhou, Barry B Snider
Index: J. Org. Chem. 73(20) , 8049-56, (2008)
Full Text: HTML
Abstract
Mander reductive alkylation of methyl 2-methoxybenzoate with prenyl bromide and hydrolysis of the enol ether afforded methyl 6-oxo-1-prenyl-2-cyclohexenecarboxylate. This was converted in five steps (reduction of the ketone, saponification, iodolactonization, ozonolysis, and intramolecular aldol reaction) to a spiro lactone cyclopentenal. An efficient first synthesis of (+/-)-vibralactone was completed by retro-iodolactonization with activated Zn, formation of the beta-lactone (vibralactone C), and reduction of the aldehyde. Except for the novel use of an iodolactone to protect both the prenyl double bond and carboxylic acid, no protecting groups were used. A similar sequence starting with asymmetric reductive alkylation of the (2S)-2-methoxymethoxymethylpyrrolidine amide of 2-methoxybenzoic acid with prenyl bromide afforded (-)-vibralactone confirming the absolute stereochemical assignment that was based on computational methods.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
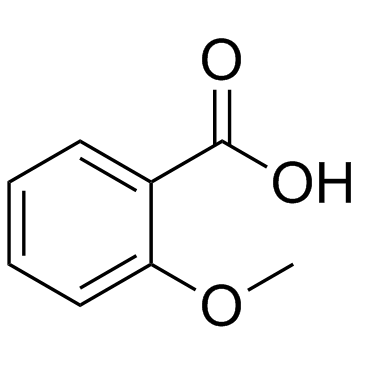 |
2-Methoxybenzoic acid
CAS:579-75-9 |
C8H8O3 |
|
Calculating virtual log P in the alkane/water system (log P(...
2005-05-05 [J. Med. Chem. 48 , 3269-79, (2005)] |
|
Endogenous salicylic acid accumulation is required for chill...
2014-10-01 [Planta 240(4) , 687-700, (2014)] |
|
Purification and properties of gentisate 1,2-dioxygenase fro...
1975-03-01 [J. Bacteriol. 121(3) , 794-9, (1975)] |
|
Effects of solute characteristics and concentration on a lyo...
1993-05-01 [Pharm. Res. 10(5) , 737-42, (1993)] |
|
[Heterocycles 39 , 47, (1994)] |