Highly-oxygenated isopimarane-type diterpenes from Orthosiphon stamineus of Indonesia and their nitric oxide inhibitory activity.
Suresh Awale, Yasuhiro Tezuka, Arjun H Banskota, I Ketut Adnyana, Shigetoshi Kadota
Index: Chem. Pharm. Bull. 51(3) , 268-75, (2003)
Full Text: HTML
Abstract
From the methanolic extract of Indonesian Orthosiphon stamineus, nine new highly-oxygenated isopimarane-type diterpenes [7-O-deacetylorthosiphol B (1), 6-hydroxyorthosiphol B (2), 3-O-deacetylorthosiphol I (3), 2-O-deacetylorthosiphol J (4), siphonols A-E (5-9)] have been isolated together with nine known diterpenes [orthosiphols H (10), K (11), M (12) and N (13); staminols A (14) and B (15); neoorthosiphols A (16) and B (17); norstaminol A (18)]. Their structures were determined based on the spectroscopic data. The isolated diterpenes inhibited nitric oxide (NO) production in lipopolysaccharide (LPS)-activated macrophage-like J774.1 cells. Compounds 4-7, 9, 10, 14, and 17 showed inhibitory activities more potent (IC(50), 10.8-25.5 microM) than a positive control N(G)-monomethyl-L-arginine (L-NMMA; IC(50), 26.0 microM).
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
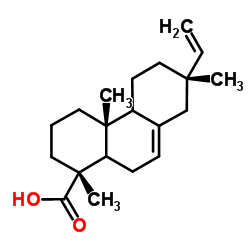 |
(5ξ,9ξ,13α)-Pimara-7,15-dien-18-oic acid
CAS:5835-26-7 |
C20H30O2 |
|
Phloroglucinols from anti-microbial deposit-resins of Austra...
2015-01-01 [Phytother Res. 29(1) , 48-58, (2015)] |
|
Spontaneous and evoked contractions are regulated by PKC-med...
2013-03-01 [Am. J. Physiol. Renal Physiol. 304(5) , F451-62, (2013)] |
|
[Analysis of main chemical composition in hydrogenated rosin...
2001-01-01 [Se Pu 19(1) , 78-81, (2001)] |
|
Influence of band 3 protein absence and skeletal structures ...
2000-06-01 [Biochim. Biophys. Acta 1466(1-2) , 125-38, (2000)] |
|
Pseudomonas reinekei sp. nov., Pseudomonas moorei sp. nov. a...
2007-05-01 [Int. J. Syst. Evol. Microbiol. 57(Pt 5) , 923-31, (2007)] |