Reaction of Lactobacillus histidine decarboxylase with L-histidine methyl ester.
T A Alston, R H Abeles
Index: Biochemistry 26(13) , 4082-5, (1987)
Full Text: HTML
Abstract
L-Histidine methyl ester inactivates histidine decarboxylase in a time-dependent manner. The possibility was considered that an irreversible reaction between enzyme and inhibitor occurs [Recsei, P. A., & Snell, E. E. (1970) Biochemistry 9, 1492-1497]. We have confirmed time-dependent inactivation by histidine methyl ester and have investigated the structure of the enzyme-inhibitor complex. Upon exposure to either 8 M guanidinium chloride or 6% trichloroacetic acid, unchanged histidine methyl ester is recovered. Formation of the complex involves Schiff base formation, most likely with the active site pyruvyl residue [Huynh, Q. K., & Snell, E. E. (1986) J. Biol. Chem. 261, 4389-4394], but does not involve additional irreversible covalent interaction between inhibitor and enzyme. Complex formation is a two-step process involving rapidly reversible formation of a loose complex and essentially irreversible formation of a tight complex. For the formation of the tight complex, Ki = 80 nM and koff = 2.5 X 10(-4) min-1. Time-dependent inhibition was also observed with L-histidine ethyl ester, L-histidinamide, and DL-3-amino-4-(4-imidazolyl)-2-butanone. No inactivation was observed with glycine methyl ester or histamine. We propose that in the catalytic reaction the carboxyl group of the substrate is in a hydrophobic region. The unfavorable interaction between the carboxylate group and the hydrophobic region facilitates decarboxylation [Crosby, J., Stone, R., & Liehard, G. E. (1970) J. Am. Chem. Soc. 92, 2891-2900]. With histidine methyl ester this unfavorable interaction is no longer present; hence, there is tight binding.(ABSTRACT TRUNCATED AT 250 WORDS)
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
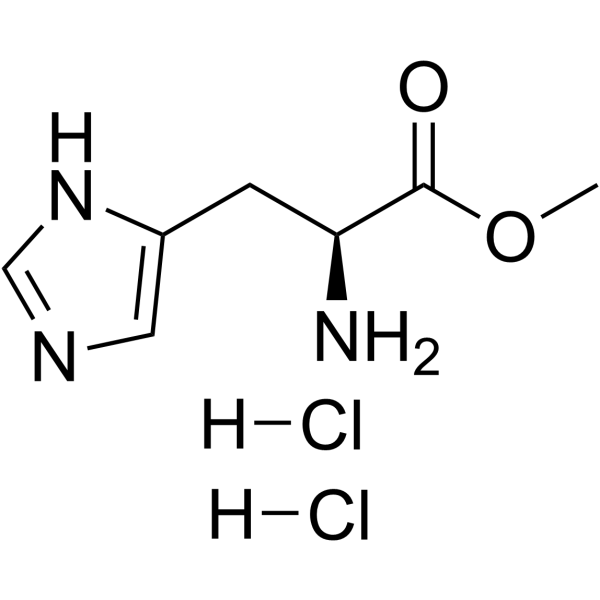 |
H-His-OMe.2HCl
CAS:7389-87-9 |
C7H13Cl2N3O2 |
|
Preparation and use of poly(hydroxyethyl methacrylate) cryog...
2013-02-01 [J. Food Sci. 78(2) , E238-43, (2013)] |
|
A proximal pair of positive charges provides the dominant li...
2012-04-01 [Biochem. J. 443(1) , 65-73, (2012)] |
|
The biological activity of hydrogen peroxide. IV. Enhancemen...
1988-03-01 [Mutat. Res. 198(1) , 233-40, (1988)] |
|
Carbonic anhydrase activators: gold nanoparticles coated wit...
2011-03-10 [J. Med. Chem. 54(5) , 1170-7, (2011)] |
|
Gas-phase structure of protonated histidine and histidine me...
2005-09-22 [J. Phys. Chem. A 109(37) , 8329-35, (2005)] |