Integrin alphaIIbbeta3:ligand interactions are linked to binding-site remodeling.
Roy R Hantgan, Mary C Stahle, John H Connor, David A Horita, Mattia Rocco, Mary A McLane, Sergiy Yakovlev, Leonid Medved
Index: Protein Sci. 15 , 1893-1906, (2006)
Full Text: HTML
Abstract
This study tested the hypothesis that high-affinity binding of macromolecular ligands to the alphaIIbbeta3 integrin is tightly coupled to binding-site remodeling, an induced-fit process that shifts a conformational equilibrium from a resting toward an open receptor. Interactions between alphaIIbbeta3 and two model ligands-echistatin, a 6-kDa recombinant protein with an RGD integrin-targeting sequence, and fibrinogen's gamma-module, a 30-kDa recombinant protein with a KQAGDV integrin binding site-were measured by sedimentation velocity, fluorescence anisotropy, and a solid-phase binding assay, and modeled by molecular graphics. Studying echistatin variants (R24A, R24K, D26A, D26E, D27W, D27F), we found that electrostatic contacts with charged residues at the alphaIIb/beta3 interface, rather than nonpolar contacts, perturb the conformation of the resting integrin. Aspartate 26, which interacts with the nearby MIDAS cation, was essential for binding, as D26A and D26E were inactive. In contrast, R24K was fully and R24A partly active, indicating that the positively charged arginine 24 contributes to, but is not required for, integrin recognition. Moreover, we demonstrated that priming--i.e., ectodomain conformational changes and oligomerization induced by incubation at 35 degrees C with the ligand-mimetic peptide cHarGD--promotes complex formation with fibrinogen's gamma-module. We also observed that the gamma-module's flexible carboxy terminus was not required for alphaIIbbeta3 integrin binding. Our studies differentiate priming ligands, which bind to the resting receptor and perturb its conformation, from regulated ligands, where binding-site remodeling must first occur. Echistatin's binding energy is sufficient to rearrange the subunit interface, but regulated ligands like fibrinogen must rely on priming to overcome conformational barriers.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
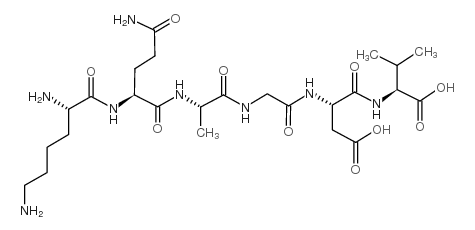 |
H-Lys-Gln-Ala-Gly-Asp-Val-OH
CAS:80755-87-9 |
C25H44N8O10 |
|
Evaluation of various silicon-and boron-containing compounds...
2014-01-01 [Eur. J. Mass Spectrom. (Chichester, Eng.) 20(4) , 337-44, (2014)] |
|
Cell adhesion peptides alter smooth muscle cell adhesion, pr...
2002-04-01 [J. Biomed. Mater. Res. 60 , 86-93, (2002)] |
|
Platelets may inhibit leucotriene biosynthesis by human neut...
2003-04-01 [Br. J. Haematol. 121 , 341-348, (2003)] |
|
The peptide Glu-His-Ile-Pro-Ala binds fibrinogen and inhibit...
1991-10-01 [Proc. Soc. Exp. Biol. Med. 198 , 649-655, (1991)] |
|
Integrins.
1991-01-01 [J. Clin. Invest. 87 , 1, (1991)] |