Antioxidant activity of 5-alkoxymethyl-6-chromanols.
M Chen, I Kohar, P T Southwell-Keely
Index: Redox Rep. 6(3) , 194-6, (2001)
Full Text: HTML
Abstract
The 5-alkoxymethyl-2,2,7,8-tetramethyl-6-chromanols (II) are excellent antioxidants against autoxidising safflower oil (ASO), although not as good as 2,2,5,7,8-pentamethyl-6-chromanol (I), the model compound of alpha-tocopherol. The aim of this work was to determine whether the rate of reaction of (II) with the radicals diphenylpicrylhydrazyl (DPP*) and galvinoxyl (ArO*) was directly proportional to their antioxidant activity against ASO. Compounds (II) reacted faster with DPP* than with ArO* but, in each case, slower than compound (I). The rates of reaction of I and II with both radicals followed the order I > II (R = H) > II (R = CH3) > II (R = other alkyls) and were directly proportional to their antioxidant activity against ASO.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
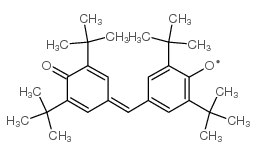 |
Galvinoxyl Free Radical
CAS:2370-18-5 |
C29H41O2 |
|
Electron-transfer mechanism in radical-scavenging reactions ...
2005-02-21 [Org. Biomol. Chem. 3(4) , 626-9, (2005)] |
|
An on-line normal-phase high performance liquid chromatograp...
2009-10-23 [J. Chromatogr. A. 1216(43) , 7268-74, (2009)] |
|
Dimerization of organic free radicals in solution. 1. Temper...
2002-04-01 [Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 58(6) , 1219-26, (2002)] |
|
Characterization of the radical-scavenging reaction of 2-O-s...
2006-04-01 [Biol. Pharm. Bull. 29(4) , 766-71, (2006)] |
|
Determination of reactions between free radicals and selecte...
2009-01-01 [Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 71(5) , 1638-43, (2009)] |