One-pot synthesis of 1,3,5-triazine derivatives via controlled cross-cyclotrimerization of nitriles: a mechanism approach.
Antonio Herrera, Alberto Riaño, Ramón Moreno, Bárbara Caso, Zulay D Pardo, Israel Fernández, Elena Sáez, Dolores Molero, Angel Sánchez-Vázquez, Roberto Martínez-Alvarez
Index: J. Org. Chem. 79(15) , 7012-24, (2014)
Full Text: HTML
Abstract
The reaction of equimolecular amounts of a nitrile and triflic anhydride or triflic acid at low temperature produces an intermediate nitrilium salt that subsequently reacts with 2 equiv of a different nitrile at higher temperature to form 2,4-disusbstituted-6-substituted 1,3,5-triazines in moderate to good yields. This synthetic procedure has also been applied to the preparation of a 1,3,5-triazine having three different substituents. The results are explained in terms of a mechanism based on the relative stability of the intermediate nitrilium salts that are formed through a reversible pathway. The formation of a substituted isoquinoline using benzyl cyanide as the second nitrile supports the postulated mechanism as well as the structure of derivatives of the proposed intermediate when the reaction is carried out in the presence of different nucleophiles other than nitriles. Theoretical calculations and the monitoring of the reaction using (1)H and (13)C NMR spectroscopy are in agreement with the proposed mechanism pathway.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
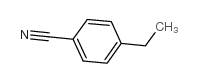 |
4-Ethylbenzonitrile
CAS:25309-65-3 |
C9H9N |
|
Rational design of novel CYP2A6 inhibitors.
2014-12-01 [Bioorg. Med. Chem. 22(23) , 6655-64, (2015)] |
|
Visible light-promoted metal-free C-H activation: diarylketo...
2013-11-20 [J. Am. Chem. Soc. 135(46) , 17494-500, (2013)] |
|
Alkylphenols and arylnitriles in a biologically active neutr...
[Phytochemistry 10(5) , 1135-1140, (1971)] |
|
1-Cyano-1, 3-butadienes. II. Carbon Structure of the Adduct ...
[J. Am. Chem. Soc. 71(3) , 1057-1058, (1949)] |
|
Arrhenius parameters for rearrangements of the neophyl, 1-in...
[J. Am. Chem. Soc. 106(14) , 3964-3967, (1984)] |