Regioselectivity in beta-galactosidase-catalyzed transglycosylation for the enzymatic assembly of D-galactosyl-D-mannose.
Mariko Miyasato, Katsumi Ajisaka
Index: Biosci. Biotechnol. Biochem. 68(10) , 2086-90, (2004)
Full Text: HTML
Abstract
The regioselectivity of beta-galactosidase derived from Bacillus circulans ATCC 31382 (beta-1,3-galactosidase) in transgalactosylation reactions using D-mannose as an acceptor was investigated. This D-mannose associated regioselectivity was found to be different from reactions using either GlcNAc or GalNAc as acceptors, not only for beta-1,3-galactosidase but also for beta-galactosidases of different origins. The relative hydrolysis rate of Gal beta-pNP and D-galactosyl-D-mannoses, of various linkages, was also measured in the presence of beta-1,3-galactosidase and was found to correlate well with the ratio of disaccharides formed by transglycosylation. The unexpected regioselectivity using D-mannose can therefore be explained by an anomalous specificity in the hydrolysis reaction. By utilizing the identified characteristics of both regioselectivity and hydrolysis specificity using D-mannose, an efficient method for enzymatic synthesis of beta-1,3-, beta-1,4- and beta-1,6-linked D-galactosyl-D-mannose was subsequently established.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
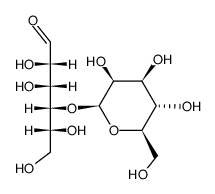 |
4-O-BETA-GALACTOPYRANOSYL-D-MANNOPYRANOSE
CAS:50468-56-9 |
C12H22O11 |
|
The nondigestible disaccharide epilactose increases paracell...
2010-02-10 [J. Agric. Food Chem. 58(3) , 1927-32, (2010)] |
|
Recent advances on physiological functions and biotechnologi...
2013-03-01 [Appl. Microbiol. Biotechnol. 97(5) , 1821-7, (2013)] |
|
Biochemical characterization of a thermophilic cellobiose 2-...
2011-01-01 [Biosci. Biotechnol. Biochem. 75 , 2162-2168, (2011)] |
|
Prebiotic properties of epilactose.
2008-12-01 [J. Dairy Sci. 91(12) , 4518-26, (2008)] |
|
Effects of epilactose on calcium absorption and serum lipid ...
2008-11-12 [J. Agric. Food Chem. 56(21) , 10340-5, (2008)] |