| Structure | Name/CAS No. | Articles |
|---|---|---|
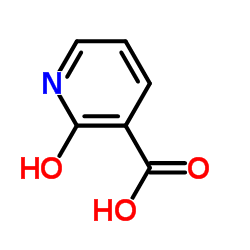 |
2-Hydroxy acid
CAS:609-71-2 |
|
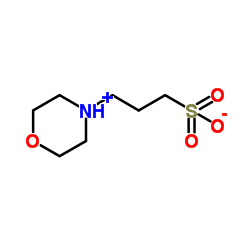 |
MOPS
CAS:1132-61-2 |