| Structure | Name/CAS No. | Articles |
|---|---|---|
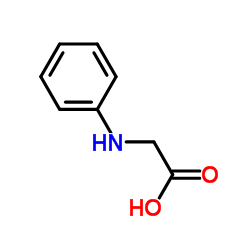 |
N-Phenylglycine
CAS:103-01-5 |
Roger Hanselmann, Jiacheng Zhou, Philip Ma, Pat N Confalone
Index: J. Org. Chem. 68(22) , 8739-41, (2003)
Full Text: HTML
Isoxazolidines have been synthesized in diastereomeric excess up to 94% via a MgBr2-induced chelation-controlled 1,3-dipolar cycloaddition reaction with N-hydroxyphenylglycinol as a chiral auxiliary. The diastereomerically pure isoxazolidines were further transformed into cyclic and acyclic beta-amino acid derivatives.
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
 |
N-Phenylglycine
CAS:103-01-5 |
C8H9NO2 |
|
Analyte-induced photoreduction method for visual and colorim...
2015-06-16 [Anal. Chim. Acta 879 , 111-7, (2015)] |
|
Notch2 controls prolactin and insulin-like growth factor bin...
2014-01-01 [PLoS ONE 9(11) , e112723, (2014)] |
|
Notch signaling governs phenotypic modulation of smooth musc...
2014-11-01 [Vascul. Pharmacol. 63(2) , 88-96, (2015)] |
|
Asymmetric strecker synthesis of alpha-amino acids via a cry...
2001-04-19 [Org. Lett. 3(8) , 1121-4, (2001)] |
|
Oncogenic deregulation of NKL homeobox gene MSX1 in mantle c...
2014-08-01 [Leuk. Lymphoma 55(8) , 1893-903, (2014)] |
Home | MSDS/SDS Database Search | Journals | Product Classification | Biologically Active Compounds | Selling Leads | About Us | Disclaimer
Copyright © 2026 ChemSrc All Rights Reserved