Interaction of the human DnaJ homologue, HSJ1b with the 90 kDa heat shock protein, Hsp90.
T Schnaider, C Soti, M E Cheetham, Y Miyata, I Yahara, P Csermely
Index: Life Sci. 67(12) , 1455-65, (2000)
Full Text: HTML
Abstract
The 90 kDa heat shock protein (Hsp90) is a major cytoplasmic molecular chaperone associating with numerous other proteins. Both genetic and in vitro refolding experiments using reticulocyte lysate have suggested a functional interaction of Hsp90 with yeast human homologues of E. coli DnaJ. Here we present direct evidence using surface plasmon resonance that Hsp90 and the human DnaJ homologue, HSJ1b, bind to each other. We also show that Hsp90 and HSJ1b transfer alpha-lactalbumin to each other in an ATP-dependent manner. The two chaperones have additive effects in preventing rhodanese aggregation.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
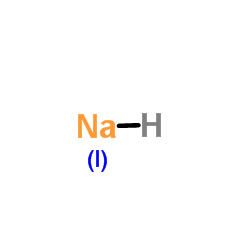 |
α-Lactalbumin
CAS:9051-29-0 |
HNa |
|
Application of circular dichroism and magnetic circular dich...
2015-03-01 [Int. J. Pharm. 480(1-2) , 84-91, (2015)] |
|
Peroxisome proliferator-activated receptor-gamma activation ...
2009-09-01 [J. Dairy Sci. 92 , 4276-89, (2009)] |
|
Why people become psychiatric nurses.
1990-01-01 [Nursing (Lond.) 4(10) , 32-4, (1990)] |
|
Experimental protein mixture for validating tandem mass spec...
2002-01-01 [OMICS 6(2) , 207-12, (2002)] |
|
Isolation and analysis of restriction endonuclease digestive...
1992-03-01 [J. Clin. Microbiol. 25(8) , 1511-5, (1987)] |