Biotechnology Letters
2003-03-01
Improving lipase-catalyzed enantioselective ammonolysis of phenylglycine methyl ester in organic solvent by in situ racemization.
Wei Du, Minhua Zong, Yong Guo, Dehua Liu
Index: Biotechnol. Lett. 25(6) , 461-4, (2003)
Full Text: HTML
Abstract
Lipase-catalyzed enantioselective ammonolysis of phenylglycine methyl ester was processed by in situ racemization with ammonium carbamate as the acyl acceptor. Using 1 mM benzaldehyde or 0.6 mM chloropyridoxal as the racemizing catalyst, 80% substrate conversion with an enantiomeric excess of the product of 95% were achieved at 20 degrees C after 7 h reaction.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
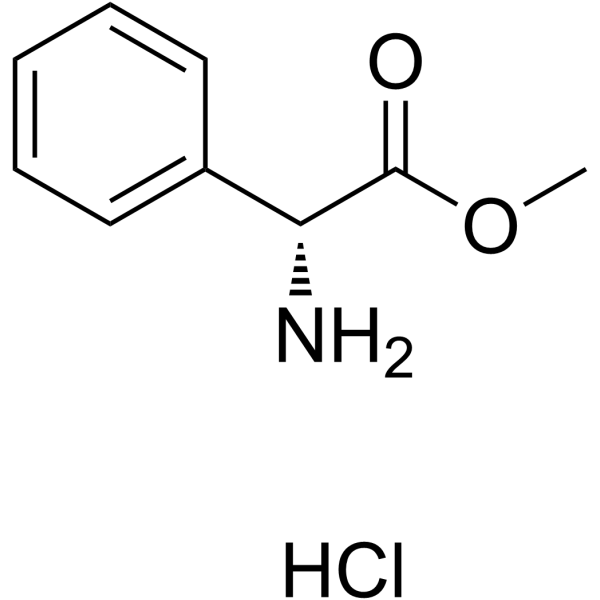 |
H-D-Phg-OMe.HCl
CAS:19883-41-1 |
C9H12ClNO2 |
Related Articles:
More...
|
Lipase-catalysed enantioselective ammonolysis of phenylglyci...
2003-10-01 [Biotechnol. Appl. Biochem. 38(Pt 2) , 107-10, (2003)] |
|
Kinetics of ampicillin synthesis catalyzed by penicillin acy...
2000-12-01 [Biochemistry. (Mosc.) 65(12) , 1367-75, (2000)] |
|
Enhanced enzymatic synthesis of a semi-synthetic cephalospri...
2003-07-01 [Biotechnol. Lett. 25(14) , 1195-8, (2003)] |
|
Life cycle assessment of a coupled solar photocatalytic-biol...
2006-11-01 [Water Res. 40(19) , 3533-40, (2006)] |
|
Increasing the synthetic performance of penicillin acylase P...
2004-07-01 [Protein Eng. Des. Sel. 17(7) , 571-9, (2004)] |