| Structure | Name/CAS No. | Articles |
|---|---|---|
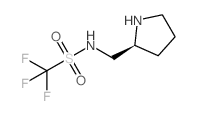 |
(S)-1,1,1-TRIFLUORO-N-(PYRROLIDIN-2-YLMETHYL)METHANESULFONAMIDE
CAS:782495-18-5 |
|
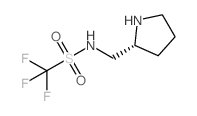 |
(R)-1,1,1-TRIFLUORO-N-(PYRROLIDIN-2-YLMETHYL)METHANESULFONAMIDE
CAS:1186049-30-8 |