1H-cyclopenta[b]benzofuran lignans from Aglaia species inhibit cell proliferation and alter cell cycle distribution in human monocytic leukemia cell lines.
F I Bohnenstengel, K G Steube, C Meyer, H Quentmeier, B W Nugroho, P Proksch
Index: Z. Naturforsch., C, J. Biosci. 54(12) , 1075-83, (1999)
Full Text: HTML
Abstract
Thirteen naturally occurring 1H-cyclopenta[b]benzofuran lignans of the rocaglamide type as well as one naturally occurring aglain congener all of them isolated from three Aglaia species (Aglaia duperreana, A. oligophylla and A. spectabilis) collected in Vietnam were studied for their antiproliferative effects using the human monocytic leukemia cell lines MONO-MAC-1 and MONO-MAC-6. Only rocaglamide type compounds showed significant inhibition of [3H-]thymidine incorporation and the most active compound didesmethylrocaglamide inhibited cell growth in a similar concentration range as the well-known anticancer drug vinblastine sulfate. Detailed structure-activity analysis indicated that the OH-group at C-8b which is a common structural feature of most naturally occurring rocaglamide compounds is essential for the described antiproliferative activity since replacement of this group by methylation led to a complete loss of the inhibitory activity for the resulting derivative. Rocaglamide derivatives rapidly inhibited DNA as well as protein biosynthesis of MONO-MAC-6 cells at concentrations well below those of actinomycin D or cycloheximide which were used as positive controls in the respective experiments. Didesmethylrocaglamide was furthermore able to induce growth arrest of MONO-MAC-1 cells in the G2/M and probably G0/G1-phase of the cell cycle with no morphological indication of cellular damage. Our data suggests that 1H-cyclopenta[b]benzofuran lignans of the rocaglamide type act primarily by a cytostatic mechanism.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
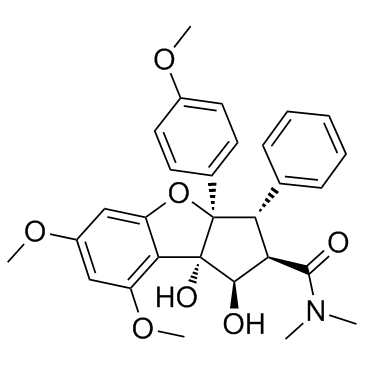 |
Rocaglamide
CAS:84573-16-0 |
C29H31NO7 |
|
Inhibitors of Translation Targeting Eukaryotic Translation I...
2012-01-01 [Meth. Enzymol. 511 , 437-61, (2012)] |
|
Rocaglamide sensitizes leukemic T cells to activation-induce...
2009-09-01 [Cell Death Differ. 16(9) , 1289-99, (2009)] |
|
Cyclorocaglamide, the first bridged cyclopentatetrahydrobenz...
2003-01-01 [J. Nat. Prod. 66(1) , 80-5, (2003)] |
|
Insecticidal rocaglamide derivatives from Aglaia spectabilis...
2000-08-01 [Phytochemistry 54(8) , 731-6, (2000)] |
|
Rocaglamide and a XIAP inhibitor cooperatively sensitize TRA...
2012-08-15 [Int. J. Cancer 131(4) , 1003-8, (2012)] |