Modification of silk fibroin using diazonium coupling chemistry and the effects on hMSC proliferation and differentiation.
Amanda R Murphy, Peter St John, David L Kaplan
Index: Biomaterials 29(19) , 2829-38, (2008)
Full Text: HTML
Abstract
A simple chemical modification method using diazonium coupling chemistry was developed to tailor the structure and hydrophilicity of silk fibroin protein. The extent of modification using several aniline derivatives was characterized using UV-vis and 1H NMR spectroscopies, and the resulting protein structure was analyzed with ATR-FTIR spectroscopy. Introduction of hydrophobic functional groups facilitated rapid conversion of the protein from a random coil to a beta-sheet structure, while addition of hydrophilic groups inhibited this process. hMSCs were grown on these modified silks to assess the biocompatibility of these materials. The hydrophilicity of the silk derivatives was found to affect the growth rate and morphology, but hMSCs were able to attach, proliferate and differentiate into an osteogenic lineage on all of the silk derivatives.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
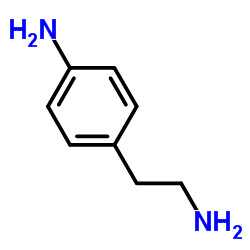 |
p-aminophenethylamine
CAS:13472-00-9 |
C8H12N2 |
|
Virtual screening identification of nonfolate compounds, inc...
2011-01-13 [J. Med. Chem. 54 , 211-221, (2011)] |
|
Efficient strategy for the selective determination of dopami...
2016-03-01 [J. Sep. Sci. 39 , 895-903, (2016)] |
|
A functional carbohydrate chip platform for analysis of carb...
2010-05-28 [Nanotechnology 21(21) , 215101, (2010)] |
|
Controlled release of cytokines using silk-biomaterials for ...
2015-12-01 [Biomaterials 73 , 272-83, (2015)] |
|
Monoamine oxidase inhibitory effects of some 4-aminophenethy...
1994-04-20 [Biochem. Pharmacol. 47(8) , 1365-71, (1994)] |