Kinetic disposition of lorazepam with focus on the glucuronidation capacity, transplacental transfer in parturients and racemization in biological samples.
Olga Papini, Sergio Pereira da Cunha, Angelo do Carmo da Silva Mathes, Carlo Bertucci, Elaine Christine Dantas Moisés, Luciana de Barros Duarte, Ricardo de Carvalho Cavalli, Vera Lucia Lanchote
Index: J. Pharm. Biomed. Anal. 40(2) , 397-403, (2006)
Full Text: HTML
Abstract
The present study investigates the kinetic disposition with focus on the racemization, glucuronidation capacity and the transplacental transfer of lorazepam in term parturients during labor. The study was conducted on 10 healthy parturients aged 18-37 years with a gestational age of 36-40.1 weeks, treated with a single oral dose of 2 mg racemic lorazepam 2-9 h before delivery. Maternal venous blood and urine samples were obtained over a 0-48 h interval and the umbilical cord sample was obtained immediately after clamping. Lorazepam enantiomers were determined in plasma and urine samples by LC-MS/MS using a Chiralcel OD-R column. In vitro racemization of lorazepam required the calculation of the pharmacokinetic parameters as isomeric mixtures. The data were fitted to two-compartment model and the pharmacokinetic parameters are reported as means (95% CI): t(1/2a) 3.2h (2.6-3.7 h), K(a) 0.23 h(-1) (0.19-0.28 h(-1)), t(1/2) 10.4h (9.4-11.3h), beta 0.068 h(-1) (0.061-0.075h(-1)), AUC(0-infinity) 175.3(ngh)/ml (145.7-204.8(ngh)/ml), Cl/F 2.6 ml/(minkg) (2.3-2.9 ml/(minkg)), Vd/F178.8l (146.5-211.1l), Fel 0.3% (0.1-0.5%), and Cl(R) 0.010 ml/(minkg) (0.005-0.015 ml/(minkg)). Placental transfer of lorazepam evaluated as the ratio of vein umbilical/maternal vein plasma concentrations, obtained as an isomeric mixture, was 0.73 (0.52-0.94). Pregnancy changes the pharmacokinetics of lorazepam, with an increase in the apparent distribution volume, an increase in apparent oral clearance, and a reduction of elimination half-life. The increase in oral clearance may indicate an increase in glucuronidation capacity, with a possible reduction in the plasma concentrations of drugs depending on glucuronidation capacity as the major metabolic pathway.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
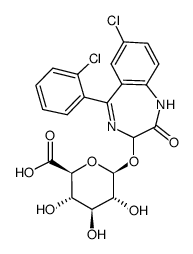 |
Lorazepam glucuronide
CAS:32781-79-6 |
C21H18Cl2N2O8 |
|
Commentary on: Dou C, Bournique J, Zinda M, Gnezda M, Nally ...
2002-03-01 [J. Forensic Sci. 47(2) , 427-8, (2002)] |
|
Population pharmacokinetics of lorazepam and midazolam and t...
2005-02-01 [Am. J. Kidney Dis. 45(2) , 360-71, (2005)] |
|
Biotransformation and excretion of lorazepam in patients wit...
1976-12-01 [Br. J. Clin. Pharmacol. 3(6) , 1033-9, (1976)] |
|
Analysis of lorazepam and its 30-glucuronide in human urine ...
2006-01-01 [J. Sep. Sci. 29(1) , 153-63, (2006)] |
|
Quantitative assay of lorazepam and its metabolite glucuroni...
2006-02-13 [J. Pharm. Biomed. Anal. 40(2) , 389-96, (2006)] |