Colorimetric method for determining Pb2+ ions in water enhanced with non-precious-metal nanoparticles.
Jeffrey Yan, Erik M Indra
Index: Anal. Chem. 84(14) , 6122-7, (2012)
Full Text: HTML
Abstract
Sulfur anions and their derivatives have long been recognized for their high selectivity and reactivity toward Pb(2+) ions and formation of highly absorptive yet water-insoluble compounds with both acid and base media. This phenomenon has been used for qualitative analysis of lead ions in water. We demonstrate a new method to quantitatively determine the Pb(2+) concentration in the range of 0.5-500 ppm in water using colorimetric measurement, based on forming "soluble" lead sulfide in water enhanced with non-precious-metal nanoparticles. This method has inherent high selectivity for lead over other alkali-metal and alkaline-earth-metal ions. The colorimetric measurements of the absorptive solutions provide accurate determination of the lead concentration in water comparable to that measured using inductively coupled plasma mass spectrometry. To our knowledge, this is the simplest, lowest cost, and easiest-to-use method for detecting and determining the lead concentration in water.
Related Compounds
| Structure | Name/CAS No. | Molecular Formula | Articles |
|---|---|---|---|
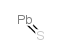 |
Lead(II) sulfide
CAS:1314-87-0 |
PbS |
|
Synthesis and spectral studies on Pb(II) dithiocarbamate com...
2012-11-01 [Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 97 , 575-81, (2012)] |
|
Harvesting solar energy by means of charge-separating nanocr...
2012-09-01 [J. Vis. Exp. (66) , e4296, (2012)] |
|
Panchromatic quantum-dot-sensitized solar cells based on a p...
2013-04-01 [ChemSusChem 6(4) , 687-92, (2013)] |
|
Effect of weathering product assemblages on Pb bioaccessibil...
2013-11-01 [Environ. Sci. Pollut. Res. Int. 20(11) , 7699-710, (2013)] |
|
Microwave-assisted cation exchange toward synthesis of near-...
2013-09-07 [Nanoscale 5(17) , 7800-4, (2013)] |